Werden Sie papierlos – Batch-Fertigung in Echtzeit mit eBMR eBMR
AmpleLogic eBMR/MES digitalisiert die Chargenfertigung mit Echtzeitausführung in der Werkstatt, Geräteintegration und Überprüfung nach Ausnahmebedingungen – wodurch die Chargenfreigabezeit um 50 % verkürzt wird und gleichzeitig die vollständige 21 CFR Part 11-Konformität erhalten bleibt.
50 %
Schnellere Chargenfreigabe
Null
Papier in der Werkstatt
99 %
Right-First-Time-Rate
10x
Schnellere Bereitstellung
Warum Papier-Chargenaufzeichnungen die Compliance bei der Pharmaherstellung behindern
Manuelle Chargenprotokolle und veraltete MES-Systeme schaffen systemische GMP-Risiken im gesamten Produktionsbetrieb – von Compliance-Gefährdungen bis hin zu Produktionsverzögerungen.
Risiko menschlicher Fehler und Manipulation
Manuelle Papieraufzeichnungen sind anfällig für Übertragungsfehler, fehlende Eingaben und Datenmanipulation – Pharma-MES-Software und elektronische Chargenfertigung eliminieren diese Risiken durch Automatisierung der Chargenfertigung.
Fehleranfällige Batch-Daten
Auf Papier aufgezeichnete Produkt- und Chargendaten verfügen nicht über eine Produktionsüberwachung in Echtzeit, was zu Spezifikationsfehlern und Nacharbeiten führt, die durch pharmazeutische Fertigungssoftware verhindert werden.
Mangelnde Echtzeit-Sichtbarkeit
Papierbasierte Systeme liefern keinen Live-Produktionsstatus – eine digitale Fertigungsplattform mit Echtzeit-Produktionsüberwachung und EE-Überwachung ersetzt verzögerte Berichte und Rundgänge.
Anspruchsvolle Audits und Inspektionen
Das Abrufen, Zusammenstellen und Überprüfen von Papierunterlagen bei FDA/MHRA-Inspektionen ist zeitaufwändig – GxP-konforme MES- und GMP-Fertigungssoftware sorgen für eine sofortige Auditbereitschaft.
Compliance- und Sicherheitslücken
Auf dem Papier fehlen robuste Zugriffskontrollen, manipulationssichere Prüfprotokolle und elektronische Signaturen, die gemäß 21 CFR Teil 11 erforderlich sind – diese MES-Plattform für Biowissenschaften schließt alle Compliance-Lücken.
Begrenzte Zusammenarbeit und Verzögerungen
Sequentielle Papierübergaben zwischen den Abteilungen verlangsamen die Batch-Ausführung – automatisierte Pharma-Regulierungs-Workflows und Systemfunktionen für die Shopfloor-Ausführung beseitigen Engpässe.
Unzuverlässige Materialverfolgung
Manuelle Materialprotokolle führen zu Diskrepanzen – Waagenintegrationen mit Barcode-Überprüfung sorgen für genaue Abgabeaufzeichnungen und Ertragsabgleich.
Schwierige Einhaltung von Standards
Die Aktualisierung von Papier-SOPs und Stammdatensätzen ist betrieblich kostspielig – die Rezepturverwaltung mit Low-Code-Fertigungsanwendungen vereinfacht Aktualisierungen über Produkte und Standorte hinweg.
Komplettes elektronisches Chargenfertigungsprotokoll und MES – eine GMP-konforme Plattform
Von der Master-Batch-Rezepterstellung bis hin zur Batch-Ausführung, Qualitätsprüfung und Freigabe – jedes eBMR-Modul arbeitet auf einer einzigen GMP-Datenschicht zusammen.
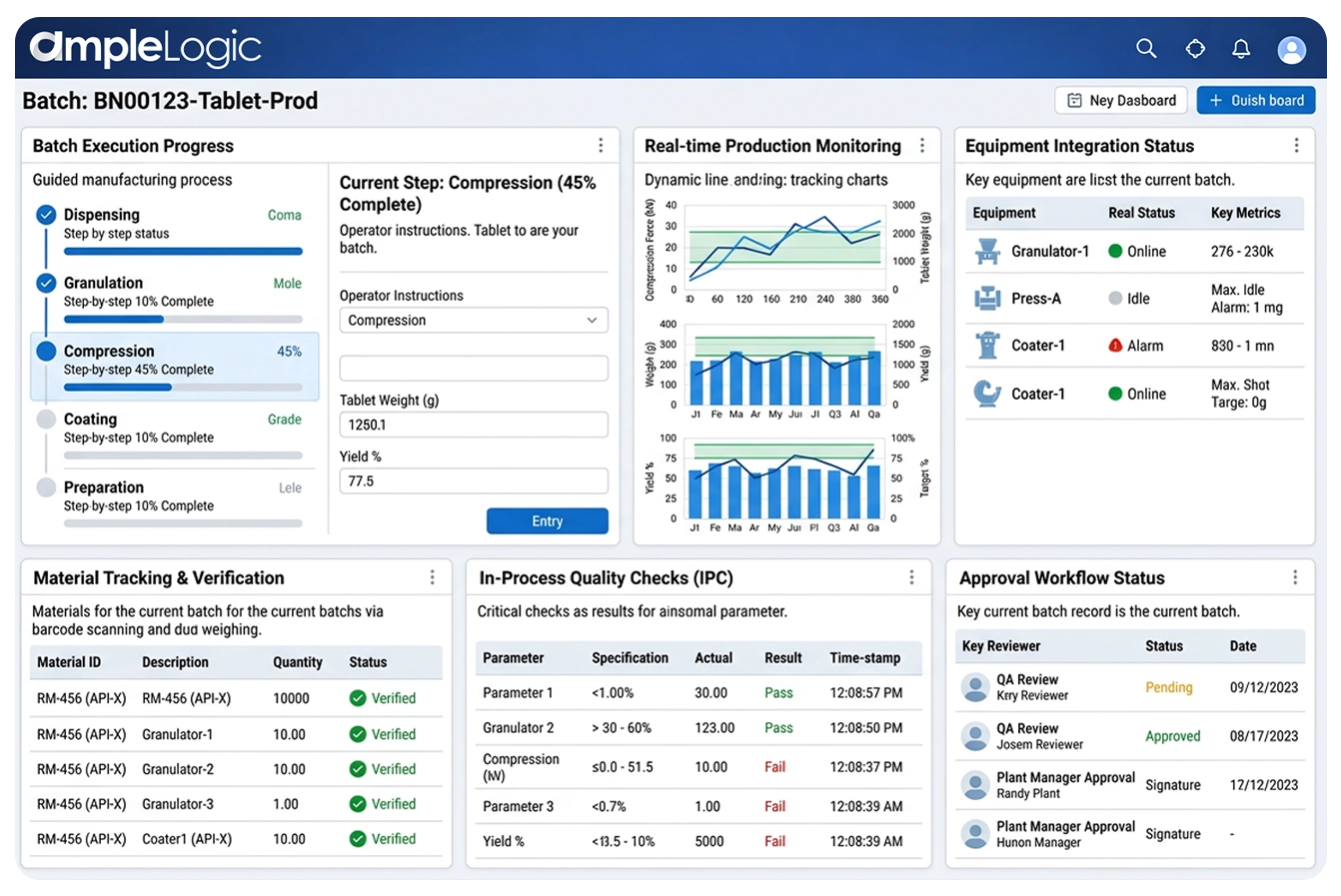
Ausführung und Produktionsüberwachung in Echtzeit
Shopfloor-Execution-System mit Echtzeit-Produktionsüberwachung, Schritt-für-Schritt-geführten Prozessen, Live-Produktions-Dashboards, EE-Überwachung und automatisierter Datenerfassung von Geräten auf dieser Pharma-MES-Software.
Leistungsfähigkeit für die pharmazeutische Fertigung auf Unternehmensniveau
Produktionsüberwachung, Geräteintegration und GMP-konforme Arbeitsabläufe – alle Funktionen für eine papierlose Pharmafertigung.
Datenerfassung und Produktionsüberwachung in Echtzeit
Pharma-MES-Software mit automatisierter Datenerfassung von Geräten, SPSen und Instrumenten – Echtzeit-Produktionsüberwachung und EE-Überwachung machen manuelle Transkription auf dieser digitalen Fertigungsplattform überflüssig.
Geräteintegration (SPS/HMI/SCADA)
Shopfloor-Execution-System mit direkter Integration über OPC, Historian-Datenbanken und Standard-SPS-Protokolle für Echtzeit-Prozessdaten auf dieser Smart-Factory-Plattform.
Elektronische Signaturen und Prüfprotokolle
GxP-konformes MES mit 21 CFR Part 11-konformen elektronischen Signaturen, unveränderlichen, zeitgestempelten Prüfprotokollen für jede Aktion, Änderung und Genehmigung in dieser GMP-Fertigungssoftware.
Automatisiertes Workflow-Management
Automatisierung der Chargenfertigung mit konfigurierbaren mehrstufigen Genehmigungsworkflows, automatischer Weiterleitung, Eskalation und paralleler/sequenzieller Verarbeitung in dieser Software für die pharmazeutische Herstellung.
Überprüfung durch Ausnahme
KI-gesteuerte Fertigungseinblicke heben nur Ausnahmen und Abweichungen für die QA-Überprüfung hervor und beschleunigen so die Chargenfreigabe auf dieser MES-Plattform für Biowissenschaften erheblich.
Erinnerungen und Eskalationswarnungen
Cloud-MES-Plattform mit automatisierten E-Mail-Benachrichtigungen für ausstehende Aufgaben, Batch-Genehmigungsphasen, überfällige Aktivitäten und Eskalation an das Management.
Produktetikettenmanagement
Digitale Fertigungsplattform mit zentraler Etikettenverwaltung, die Konsistenz, Versionskontrolle und Einhaltung gesetzlicher Vorschriften aller Produktetiketten gewährleistet.
Verpackungskomponenten
Pharmazeutische Fertigungssoftware, die unterschiedliche Verpackungsanforderungen pro Produkt mit konfigurierbaren Stücklisten, Komponentenverfolgung und -abgleich abdeckt.
Low-Code-Fertigungsanwendungen
Low-Code-Fertigungsanwendungen mit intuitiver Drag-and-Drop-Schnittstelle zur Anpassung von Prozessen – ermöglichen die Digitalisierung der Pharmafertigung ohne benutzerdefinierte Programmierung.
Erweiterte Produktionsplanung
Intelligente Fabrikplattform mit Industrie 4.0-Fertigungsplanungsalgorithmen, die die Ressourcenzuteilung optimieren, Umrüstzeiten minimieren und den Durchsatz maximieren.
Skalierbare Cloud-MES-Plattform
Cloud-MES-Plattform, die zu 100 % anpassbar ist – skalierbar von einer Einzellinie bis hin zur unternehmensweiten Bereitstellung an mehreren Standorten für die Anforderungen einer Biopharma-Produktionsplattform.
Vollständige Sichtbarkeit des Produktionslebenszyklus
Digitale Fertigungstransformation mit durchgängiger Transparenz und Kontrolle über den gesamten Produktionslebenszyklus von der Planung bis zur Chargenfreigabe und Archivierung.
Vereinfachte Chargenfreigabe von Arzneimitteln
Ein strukturierter End-to-End-eBMR-Ablauf, der Ihren pharmazeutischen Herstellungsprozess von der Rezepterstellung bis zur endgültigen Chargenfreigabe leitet.
Master-Setup
- Erstellung von Mastern (Produkte, Materialien, Geräte, Räume)
- Produkt- und Bühnenzuordnung
- Zuordnung von Rohstoffen zu Produkten
- Raum- und Gerätekartierung
- Sequentielle und parallele Prozessdefinition
Rezept & Terminplanung
- Erstellung von Grundrezepten (Drag-and-Drop)
- Produktionsplanung und Ressourcenzuweisung
- Chargenplanung und -ausgabe
Batch-Ausführung
- Abgabe von Rohstoffen mit Barcode-Verifizierung
- Schritt-für-Schritt-geführte Batch-Ausführung
- Geräteintegration und automatische Datenerfassung
- Prozessbegleitende Qualitätskontrollen und Testergebnisse
- Raum- und Geräteprotokolleinträge
Überprüfung und Veröffentlichung
- Ausnahmebehandlung und Abweichungsmanagement
- Elektronische Signaturen und mehrstufige Genehmigungen
- Überprüfung nach Ausnahme für eine schnellere Chargenfreigabe
- Vollständiger Audit-Trail und Batch-Genealogie
Schrittweise Migration: Vom Papier zum vollständigen eBMR
BIMS (Batch Record Issuance Management)
- Verwaltung von Masterkopien und Ausgabe von Dokumenten
- Automatisches Drucken zur Vermeidung manueller Fehler
- Mutter-Batch-, Co-Pack- und Bilayer-Logik
- Mengenmanagement (Soll & Haben)
- Kontrolle auf manuelle Aufnahmefehler, zusätzliche Seitenanfragen, Ratifizierungen
eBatch-Aufzeichnung
- Ausstellungsprozess auf Online-Workflow umgestellt
- Chargeninformationen online aufzeichnen (von der Papierkopie wechseln)
- Maßnahmen zur Bestätigung der Aufzeichnung erfolgen nur am Gerät
- Integration mit eQMS-Systemen
Vollständiges eBMR
- Wechseln Sie von der manuellen Aufzeichnung zum automatischen Ausfüllen von Daten aus Geräten
- Echtzeit-Datenintegration mit SPS, HMI und SCADA
- Komplett papierlose Fertigung
- Vollständige Konformität mit 21 CFR Part 11 / EU Annex 11
Vereinfachen Sie die Ausführung der Pharmafertigung mit elektronischen Chargenprotokollen
Speziell für Pharmaunternehmen in der GMP-Fertigung entwickelt – 100 % Compliance, automatisierte Validierung.
Geführte Prozesse
Shopfloor-Execution-System mit benutzerfreundlichen Schritt-für-Schritt-Anleitungen zur Ausführung von Fertigungsschritten – Bediener folgen intuitiven Anweisungen, um die Konsistenz dieser Pharma-MES-Software zu gewährleisten.
Produktionsüberwachung in Echtzeit
Echtzeit-Produktionsüberwachung mit animierten visuellen Hinweisen und Statussymbolen, die den Bedienern hilft, den aktuellen Aktivitätsstatus auf einen Blick zu verstehen, mit EE-Überwachung auf dieser digitalen Fertigungsplattform.
Automatische Validierungen
Automatisierung der Chargenfertigung mit Systemwarnungen bei falschen Eingaben, außerhalb der Spezifikation liegenden Werten und verpassten Schritten, um Fehler zu verhindern, bevor sie in die elektronische Chargenfertigungsaufzeichnung gelangen.
SOPs auf dem Bildschirm
GMP-Fertigungssoftware, die Standardarbeitsanweisungen direkt am Verwendungsort auf dem Bildschirm anzeigt, sodass sie während der Ausführung sofort darauf zurückgegriffen werden kann.
Aktivitätsanleitung und nächste Schritte
Smart-Factory-Plattform mit klaren Anweisungen für die nächsten Schritte bei jeder Aufgabe, damit die Bediener auf dieser MES-Plattform für die Biowissenschaften immer wissen, was als nächstes im Prozess ansteht.
Barcode- und QR-Scannen
Waagenintegrationen und integriertes Barcode-/QR-Scannen zur Materialüberprüfung, Geräteidentifizierung und fehlersicheren Dateneingabe in der Industrie 4.0-Fertigungshalle.
Nahtlos integriertes eBMR/MES in Ihrem gesamten Pharmabetrieb
Native eBMR/MES-Integration über Qualitäts-, Labor-, Schulungs-, Ausrüstungs- und Unternehmenssysteme hinweg – alles auf einer 21 CFR Part 11-konformen Plattform.
Qualitätssysteme
- eQMS (CAPA, Abweichungen, Änderungskontrolle)
- OOS/OOT-Management
- Auditmanagement
- Beschwerdebearbeitung
Labor
- LIMS (In-Process Testing, CoA)
- Stabilitätsstudien
- Umweltüberwachung (EMS)
Dokument & Schulung
- DMS (SOPs, Master Records)
- LMS (Bedienerschulung, Kompetenz)
- Elektronische Fahrtenbücher (eLogs)
Ausrüstung und Automatisierung
- SPS-/HMI-/SCADA-Integration
- OPC-Server-Konnektivität
- Historikerdatenbank
- Wiege- und Dosiersysteme
Unternehmenssysteme
- SAP/Oracle ERP
- Lagerverwaltung (WMS)
- Lieferkettenmanagement
- Finanzsysteme
Kalibrierung und Wartung
- CAPS (Kalibrierungsplanung)
- Vorbeugende Wartung
- Gerätequalifikation
- Asset-Lebenszyklus
Wie AmpleLogic eBMR/MES im Vergleich zu herkömmlichen Pharma-MES-Lösungen abschneidet
Erfahren Sie, wie AmpleLogic eBMR im Vergleich zu herkömmlichen, unternehmensweiten und automatisierungsorientierten pharmazeutischen MES-Lösungen abschneidet.
| Besonderheit | AmpleLogic | Cloud-MES | Unternehmens-MES | Automatisierung MES |
|---|---|---|---|---|
| Bereitstellungsgeschwindigkeit | Wochen (Low-Code aPaaS) | Monate (Cloud SaaS) | 6–18 Monate (On-Prem/Cloud) | 6–12 Monate (On-Prem/Cloud) |
| Anpassung | 100 % Drag-and-Drop, kein Programmieren | Vorlagenbasierte Konfiguration | Professionelle Dienstleistungen erforderlich | Erfordert Systemintegratoren |
| Rezeptverwaltung | Visueller Drag-and-Drop-Builder | Vorlagengesteuerter EBR | Rezeptgesteuerte ISA-88-Engine | Rezeptverwaltungsmodul |
| Geräteintegration | SPS/HMI/SCADA über OPC | Begrenzt (QMS-fokussiert) | Tief ISA-95 / OPC-UA | Anbietergebundenes SPS-Ökosystem |
| Qualitätssystemintegration | Native (gleiche Plattform) | Natives QMS + EBR | Integration von Drittanbietern | Integration von Drittanbietern |
| Einheitliche Plattform | Über 14 Apps auf einem einzigen aPaaS | Nur QMS + Fertigung | Nur MES (geschlossenes Ökosystem) | Nur MES (Anbieter-Ökosystem) |
| Infrastrukturkosten | Bis zu 70 % niedriger (Open-Source) | Cloud-Abonnementmodell | Hoch (Unternehmenslizenzierung) | Hoch (Plattform + Integratoren) |
| Kontinuierliche Produkt-Upgrades | Inklusive regelmäßiger Veröffentlichungen | Automatische Cloud-Updates | Versions-Upgrades (projektbasiert) | Hauptversionsveröffentlichungen |
100 % anpassbar über Low-Code
Low-Code-Fertigungsanwendungen – jeder Arbeitsablauf, jede Form und jeder Prozess ist vollständig konfigurierbar, um Ihren genauen Fertigungsanforderungen zu entsprechen, ohne dass benutzerdefinierte Programmierung in dieser Pharma-MES-Software erforderlich ist.
Drag-and-Drop-Rezept-Builder
Rezeptverwaltung mit visuellem Rezepterstellungstool, mit dem Prozessingenieure auf dieser digitalen Fertigungsplattform ohne IT-Einbindung Hauptrezepte erstellen und ändern können.
Cloud-MES-Plattform mit kontinuierlichen Upgrades
Cloud-MES-Plattform mit regelmäßigen Produktveröffentlichungen, neuen Funktionen, Aktualisierungen von Vorschriften und Leistungsverbesserungen – die in Ihrem Abonnement enthalten ist und die Transformation der digitalen Fertigung ermöglicht.
Schnellere Bereitstellung für Industrie 4.0
Smart-Factory-Plattform und Life-Science-MES-Plattform ermöglichen die Bereitstellung innerhalb von Wochen – nicht in den 6–18 Monaten, die für herkömmliche MES-Implementierungen typisch sind – und beschleunigen so die Digitalisierung der Pharmaproduktion.
Der Business Case für die papierlose pharmazeutische Herstellung
Quantifizierte Ergebnisse – Beschleunigen Sie die Chargenfreigabe, reduzieren Sie die Papierkosten und verbessern Sie die Fertigungsgenauigkeit nach dem Einsatz der AmpleLogic Electronic Batch Manufacturing Record-Software.
Schnellere Chargenfreigabe
Durch Ausnahmeprüfungen und automatisierte Arbeitsabläufe wird die Zeit für die Stapelfreigabe im Vergleich zu papierbasierten Prozessen halbiert.
Reduzierung der Papierkosten
Eliminieren Sie in Ihren Fertigungsabläufen Papierchargenprotokolle sowie Druck-, Lager- und Abrufkosten.
Weniger Dateneingabefehler
Durch automatische Validierung, Geräteintegration und geführte Prozesse werden manuelle Übertragungsfehler praktisch ausgeschlossen.
Niedrigere Infrastrukturkosten
Der Open-Source-Infrastruktur-Stack (Linux, PostgreSQL) reduziert die Ausgaben für die IT-Infrastruktur erheblich.
Kapitalrendite
Kunden erzielen in der Regel innerhalb der ersten 18 Monate nach der Bereitstellung durch betriebliche Effizienz einen drei- bis fünffachen ROI.
Schnellere Audit-Vorbereitung
Der sofortige Zugriff auf unveränderliche elektronische Aufzeichnungen mit vollständigen Prüfprotokollen beschleunigt die Inspektionsbereitschaft.
eBMR/MES-konform mit über 16 globalen Pharmaaufsichtsbehörden
Gebaut für die Einhaltung pharmazeutischer Vorschriften vom ersten Tag an – FDA 21 CFR Part 11, EU Annex 11, GAMP 5 und mehr.
Compliance-Standards
Aufsichtsbehörden
USFDA
USA
MHRA
Vereinigtes Königreich
TGA
Australien
CDSCO
Indien
Gesundheit Kanada
Kanada
MCC
Südafrika
ANVISA
Brasilien
EMEA
europäische Union
SFDA
China
MPA
Schweden
Swissmedic
Schweiz
MCAZ
Simbabwe
MEDSAFE
Neuseeland
MHLW
Japan
NAFDAC
Nigeria
KFDA
Korea
Speziell für die regulierte Fertigung entwickelt
Pharmazeutische Herstellung
- Orale feste Dosierung
- Flüssig und halbfest
- Sterile Injektionspräparate
- API-Herstellung
- Digitalisierung der Pharmaherstellung
Biopharma-Produktionsplattform
- Produktion von Biologika
- Zellkultur
- Reinigung
- Füllen und fertigstellen
- Ausführung der Biotech-Produktion
Medizinische Geräte
- Gerätemontage
- Verpackung und Etikettierung
- Sterilisationsaufzeichnungen
- DHR-Management
CDMO / CMO
- Multi-Client-Chargenaufzeichnungen
- Kampagnenherstellung
- Technologietransfer
- Schnelles Site-Onboarding
- Transformation der digitalen Fertigung
"Die Produktionsaktivitäten sind jetzt sehr einfach und stabil. Der Übergang von Papier-BMRs zu elektronischen hat uns viel Zeit gespart und Fehler reduziert."
Produktionsleiter
Mittelständisches Pharmaunternehmen
"Erleichtert die Ausstellung und Nachverfolgung von BMRs erheblich. Der papierlose Arbeitsablauf optimierte unseren gesamten Chargendokumentationsprozess."
QA-Spezialist
Unternehmensfertigung
"Einfach zu implementieren und das Support-Team hat uns durch den gesamten Prozess geführt. Die Integration in unser Dokumentenmanagementsystem funktioniert reibungslos."
IT-Direktor
Pharmaunternehmen
Sind Sie bereit, in Ihrer Werkstatt auf Papier zu verzichten?
Erfahren Sie, wie AmpleLogic eBMR/MES Ihre Serienfertigung digitalisieren und die Freigabezeit um 50 % verkürzen kann. Vereinbaren Sie noch heute eine personalisierte Demo.
Bleiben Sie in den Biowissenschaften an der Spitze
Erhalten Sie die neuesten Produktaktualisierungen, Compliance-News und Brancheneinblicke in Ihren Posteingang.