Ersetzen Sie Papier-Logbücher durch ALCOA+-konforme digitale Protokolle
AmpleLogic ist ein GMP-konformes elektronisches Logbuchsystem, das speziell für die Anforderungen der Pharma-Produktionslogbuchsoftware entwickelt wurde. Schließen Sie die betriebliche Kontextlücke zwischen Altgeräten und modernem MES durch KI-gestützte papierlose Logbücher in der Pharmaproduktion – digitalisieren Sie Beobachtungen, ohne die validierte Systemlogik zu beeinträchtigen.
100 %
Audit-Trail-Tiefe
#1
Regulierungsangleichung
100 %
Einführung der elektronischen Signatur
10x
Schnellere Bereitstellung mit KI
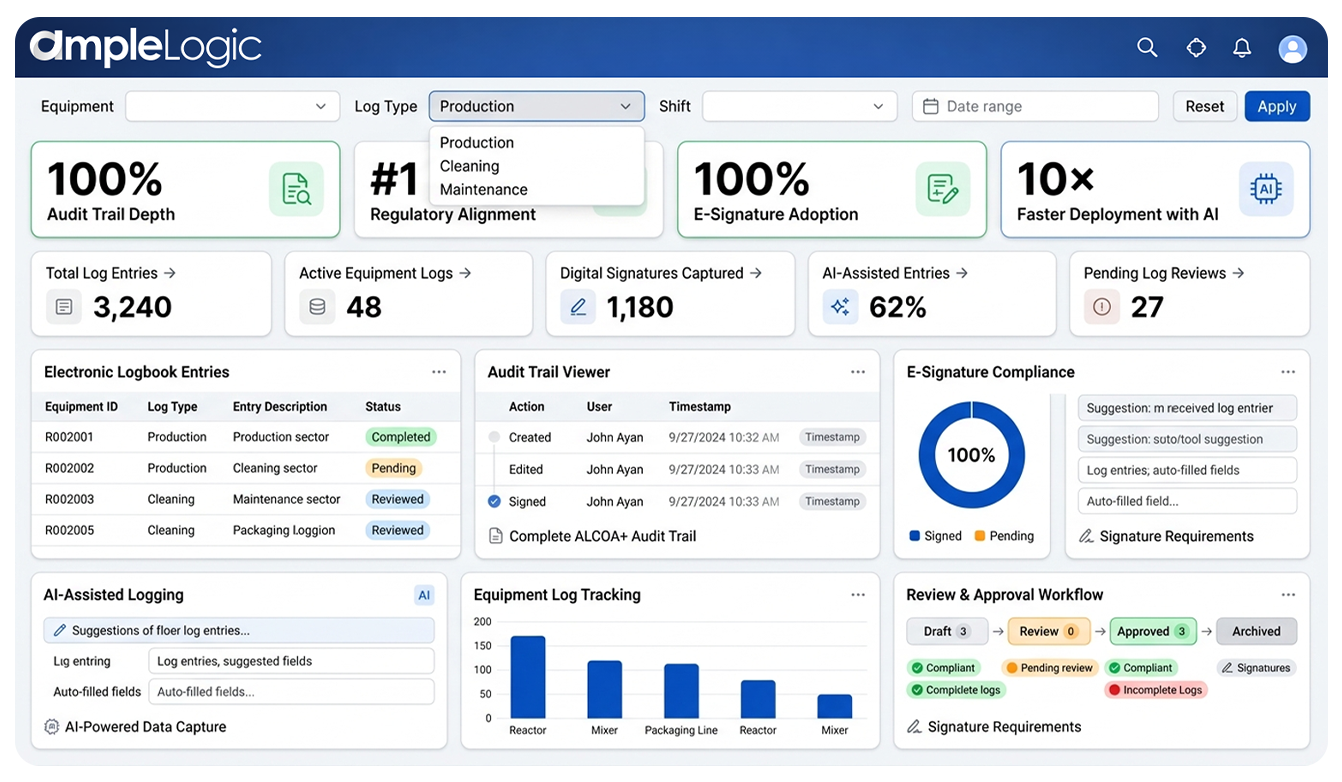
21 CFR Part 11 & EU Annex 11 konformes elektronisches Logbuch für Pharma
Die eLogbook-Architektur ist so konzipiert, dass sie die strengsten gesetzlichen Anforderungen an elektronische Aufzeichnungen und Datenintegrität erfüllt
Audit-Trail-Tiefe
Vollständige Erfassung aller Eingaben, Änderungen und Löschungen mit Zeitstempeln. ALCOA+ zuordenbare und zeitgenössische Aufzeichnungen vom ersten Tag an.
Regulierungsangleichung
Vollständig konform mit den Rahmenwerken FDA 21 CFR Part 11 und EU Annex 11. Entwickelt, um die strengsten GxP-Anforderungen weltweit zu erfüllen.
Einführung der elektronischen Signatur
Erzwungene biometrische oder MFA-Authentifizierung für alle wichtigen GxP-Genehmigungen. Unbestreitbarkeit und Signaturverknüpfung gemäß den Anforderungen von Teil 11.
Datenintegrität durch Design
Jede Aufzeichnung ist zuordenbar, lesbar, zeitgleich, original und korrekt. Vollständigkeit, Konsistenz, Ausdauer und Verfügbarkeit werden automatisch erzwungen.
eLogbook: Der flexible Gerätebetrieb und die Offline-Aufzeichnung für den Betrieb in den Biowissenschaften
eLogbook dient als kontextuelle Brücke sowohl über die automatisierte MES-Ausführung als auch über Rohdaten der Instrumentierung
MES-Ausführungsschicht
Strukturierte Transaktionsdaten und Durchsetzung von Chargenprotokollen für automatisierte Hochgeschwindigkeitslinien. Verarbeitet binäre Pass/Fail-Transaktionen und strukturierte Fertigungsabläufe.
eLogbook-Kontextschicht
eLogbuchErfasst Beobachtungsdaten, Situationsbeurteilungen und ausnahmebasierten Betriebskontext. Die flexible Dokumentationsebene über automatisierten und manuellen Fertigungsaktivitäten.
Instrumentierungsschicht
Rohdaten von SPS, SCADA und eigenständigen Altgeräten in der gesamten Anlage. Gerätemesswerte, Sensordaten und automatisierte Alarme werden in Echtzeit in das eLogbook eingespeist.
Warum MES allein für die Einhaltung der Pharma-GMP-Dokumentation nicht ausreicht
Bei Transaktionssystemen steht die Ausführungsgeschwindigkeit im Vordergrund und dabei geht häufig der wichtige menschliche Kontext der Vorgänge verloren
Indirekte tote Winkel der Ausrüstung
MES fehlt die Flexibilität, indirekte Geräte wie HLK- oder Reinwassersysteme ohne umfangreiche Anpassungen zu dokumentieren. eLogbooks erfassen diese kritischen Beobachtungen nativ.
Fehlender menschlicher Kontext
ELBs erfassen Bedienerbewertungen und Situationsbeurteilungen, bei denen es sich nicht um binäre Pass/Fail-Transaktionen handelt. Der menschliche Kontext des Betriebs bleibt für Audits und Untersuchungen erhalten.
Unstrukturierte Aktivitätslücken
Die Dokumentation unstrukturierter Aktivitäten wie Schichtübergaben und Ausnahmemeldungen wird in eLogbooks nativ unterstützt, im Gegensatz zu strukturierter MES-Logik.

Der eLogbook-Vorteil
MES vs. elektronisches Logbuch: Vergleich des Umfangs der GMP-Dokumentation
Vergleich des Dokumentationsumfangs über die Ebenen „Fertigungsausführung“ und „Logbuch“ hinweg
| Aktivitätskategorie | MES-Fokus | eLogbook-Fokus |
|---|---|---|
| Reinigung und Räumung | Pass/Fail-Zeitstempel | Visuelle Beobachtungen und spezifische Methoden |
| Versorgungsüberwachung | Automatisierte Alarme | Situative Darstellung von Abweichungen (z. B. HVAC) |
| Schichtübergänge | N/A (prozessspezifisch) | Kontinuitätsnarrativ und institutionelles Wissen |
| Kalibrierung | Referenzdatum | Verifizierungsprotokolle und manuelle Geräteprüfungen |
| Umweltüberwachung | Automatisierte Sensorprotokolle | Bedienerbeobachtungen, Abweichungsberichte, Korrekturnotizen |
| Materialhandhabung | Barcode-Scan-Ereignisse | Empfangsbedingungen, Sichtprüfungen, Lagerausnahmen |
GMP-Geräte-Logbuch-Software für Pharma, Biotechnologie und regulierte Fertigung
Vom Drag-and-Drop-Logbuch-Builder bis zur Reinraum-Logbuch-Software und Lager-Logbuch-Software – alle Funktionen sind für GxP-regulierte Umgebungen konzipiert
Echtzeit-Datenintegration
Live-Feeds von SPS, SCADA und eigenständigen Instrumenten fließen direkt in das elektronische Logbuch für die pharmazeutische Produktion. Eliminieren Sie manuelle Übertragungsfehler und stellen Sie eine zeitgleiche Datenerfassung in der gesamten Einrichtung sicher.
Drag-and-Drop-Logbuch-Builder
Geräte-Logbuch-Software für Pharma
Reinraum-Logbuch-Software
Kalibrierungs-Logbuch-Software
QC-Labor-Logbuch-Software
Logbuch zur Umweltüberwachung
Lagerlogbuch-Software
Rollenbasierte Logbücher
Checklisten und Verriegelung
Intelligente Benachrichtigungen und Warnungen
Analysen und Dashboards
QR-/Barcode-Scannen
Offline-Zugriff (native App)
Sprachgesteuerte Protokollierung
Unveränderlicher Prüfpfad
Intelligente Berichtserstellung
E-Signatur-Workflows
Elektronisches Logbuch über 4 pharmazeutische Betriebssäulen hinweg
Konsolidierte automatisierte Protokollierungsdomänen innerhalb des AmpleLogic eLogbook-Ökosystems
Bereichs- und Geräteprotokolle
- Überwachen Sie den Betrieb von Klimaanlagen, Kältemaschinen und Kesseln mit der Logbuch-Software für die Pharmaindustrie
- Automatisieren Sie die Kalibrierungslogbuchsoftware und die Aufzeichnungen zur vorbeugenden Wartung
- Verfolgen Sie den Lebenszyklus des Gerätewartungslogbuchs, Statusänderungen und Nutzung
- Erfassen Sie die Umgebungsbedingungen mithilfe des Umweltüberwachungslogbuchs für klassifizierte Bereiche
QC-Laborbetrieb
- Schließen Sie die LIMS-Lücke mit der QC-Laborlogbuchsoftware für physische Inspektionen und Desinfektion
- Verfolgen Sie Protokolle zum Säulenlebenszyklus und zur Instrumentennutzung
- Protokollieren Sie die Vorbereitung, Standardisierung und das Verfallsdatum der Reagenzien
- Dokumentieren Sie Medienvorbereitung und Wachstumsförderungstests
Produktion & Lager
- Digitalisieren Sie Dosier-, Verpackungs- und Beschichtungsprotokolle mit der Logbuchsoftware für die Pharmaproduktion
- Lagerlogbuch-Software für Materialtransport und Umweltaufzeichnungen
- Verfolgen Sie In-Prozess-Kontrollen und Line-Clearance-Aktivitäten
- Start/Stopp des Dokumentstapels, Ertragsabgleich und Ausnahmen
Qualität und Compliance
- Automatisieren Sie OOC-, Verlängerungsanfrage- und Abweichungsprotokolle
- Protokollieren Sie die Teilnahme an Schulungen und die Materialausgabe
- Verwalten Sie Narrative und Folgenabschätzungen zur Änderungskontrolle
- Verfolgen Sie CAPA-Aktionen, die mit Logbuchbeobachtungen verknüpft sind

Implementierungs-Roadmap
Legacy-Bewertung
Identifizieren Sie Papierprotokolle mit hohem Risiko und veraltete Geräte ohne digitale Ausgabe. Ordnen Sie kritische Dokumentationslücken zu und priorisieren Sie Bereiche für maximale Compliance-Auswirkungen.
Nicht aufdringliches Overlay
Stellen Sie eLogbook-Vorlagen bereit, ohne die zugrunde liegende Steuerlogik oder den validierten SPS-/SCADA-Code zu ändern. Vermeiden Sie kostspielige Neuvalidierungszyklen durch schreibgeschützte Integration.
Validierung und Schulung
Führen Sie 21 CFR Part 11-Validierungsprotokolle aus und schulen Sie Bediener in der digitalen Beobachtungserfassung. Umfangreiche IQ/OQ/PQ-Dokumentation inklusive.
Unternehmenssynergie
Integrieren Sie eLogbook-Daten mit MES, ERP, eQMS und LIMS für eine einheitliche Betriebsberichterstattung. Schaffen Sie eine zentrale Informationsquelle im gesamten Fertigungsunternehmen.
KI-gesteuerte automatische Generierung von GxP-Protokollen aus Word-Vorlagen zur Reduzierung der Dokumentationszeit
Die Low-Code-Engine von AmpleLogic wandelt Standard-Word-Vorlagen sofort in vollständig kompatible, versionierte elektronische Datensätze um – die KI-gesteuerte automatische Generierung von Protokollen aus Word-Vorlagen macht die manuelle Konfiguration vollständig überflüssig
KI-gesteuerte automatische Generierung von Protokollen aus Word-Vorlagen
Laden Sie eine beliebige vorhandene Word-Vorlage hoch. Die KI erkennt automatisch Datenstrukturen und bildet Compliance-Felder ohne manuellen Aufwand ab. Durch die KI-gesteuerte automatische Generierung von Protokollen aus Word-Vorlagen werden Papierformulare sofort in digitale E-Logbücher umgewandelt – wochenlange manuelle Konfiguration entfällt.
Automatisierte Compliance
Generierte eLogbücher entsprechen grundsätzlich den Standards FDA 21 CFR Part 11 und EU Annex 11. Dieses GMP-konforme elektronische Logbuchsystem bietet vom ersten Tag an globale Auditbereitschaft, ohne dass eine manuelle Compliance-Konfiguration erforderlich ist.
Nahtlose Versionierung
Die integrierte Nachverfolgung erfasst jede Änderung im gesamten Dokumentlebenszyklus. Bietet einen transparenten und unveränderlichen Prüfpfad mit vollständiger Rückverfolgbarkeit darüber, wer wann was geändert hat.
Schnelle Bereitstellung (10x schneller)
Erreichen Sie den Go-Live-Status 10x schneller als mit herkömmlichen Methoden. KI reduziert die Zeit, die Geschäftsteams für Konfiguration, Validierung und Vorlagenerstellung aufwenden, erheblich.
Anomalieerkennung
KI erkennt Datenausreißer, um Fehler, Compliance-Probleme oder außerhalb der Spezifikation liegende Bedingungen in Echtzeit zu identifizieren. Proaktive Warnungen verhindern Abweichungen, bevor sie eskalieren.
Vorausschauende Wartung
KI sagt Geräteausfälle anhand historischer Logbuchdaten und Sensortrends voraus. Automatisierte Warnmeldungen verhindern ungeplante Ausfallzeiten und stellen die Einhaltung von Wartungsplänen sicher.
Kostengünstige elektronische Logbuch-Alternative zu kundenspezifischen Pharma-Software-Integrationen
eLogbooks machen maßgeschneiderte Hardwareschnittstellen für jedes Teil der Werkstattausrüstung überflüssig
Benutzerdefinierte Hardwareschnittstellen für ältere Geräte
Benutzerdefinierte Hardware- und Punktintegrationen
Keine kundenspezifische Hardware; nicht-intrusives ELB
Zeitpläne für die Bereitstellung
Lange MES-Programme (Jahre)
Schnelle ELB-Einführung (Monate)
Lebenszykluswartung und Upgrades
Eng gekoppelte Systeme
Entkoppelte ELB-Architektur
Ein Weg zur digitalen Reife
Pragmatische Digitalisierung
Ersetzen Sie Papierprotokolle durch eLogbücher für sofortige Compliance-Erfolge und Datenintegrität. Erreichen Sie Auditbereitschaft, ohne validierte Systeme zu stören.
Vorlagenstandardisierung
Vereinheitlichen Sie die Dokumentation über Schichten, Standorte und Abteilungen hinweg, um den institutionellen Wissenserhalt und konsistente GxP-Praktiken weltweit sicherzustellen.
Integrierter Kontext
Synergisieren Sie eLogbook-Daten mit MES-Metriken für eine ganzheitliche 360-Grad-Ansicht der Charge. Ermöglichen Sie prädiktive Analysen und kontinuierliche Verbesserung.
Digitalisierung älterer GxP-Umgebungen mit gesetzeskonformer elektronischer Logbuch-Software
Nicht-intrusive Integration mit eigenständigen Instrumenten, SPS, SCADA-Systemen und Unternehmensanwendungen
eQMS
Automatisierte Abweichungs-, CAPA- und Änderungskontroll-Workflows verknüpft mit Logbuchbeobachtungen
LIMS
Synchronisieren Sie Laborergebnisse, Geräteprotokolle und QC-Probenverfolgung in Echtzeit
ERP & Wartung
Integrieren Sie Gerätestatus, Ersatzteile und vorbeugende Wartungspläne
eBMR / MES
Bidirektionale Batch-Kontextfreigabe zwischen Ausführungsdatensätzen und Beobachtungsprotokollen
SPS / SCADA / Datenlogger
Nichtintrusive schreibgeschützte Verbindungen über RS 232/485-, Ethernet-, API- oder OPC-Protokolle
Anwendungen von Drittanbietern
Offene API und CAMS/OPC/Link-Server-Konnektivität für jede externe Systemintegration

Integrität der Gerätelogik
Erreichen Sie die Digitalisierung, ohne den validierten SPS- oder SCADA-Code zu ändern. Durch schreibgeschützte, nicht aufdringliche Verbindungen werden kostspielige Neuvalidierungszyklen vermieden und gleichzeitig der vollständige Betriebskontext erfasst.
Branchen, die das elektronische Logbuch von AmpleLogic für die GMP-Pharmaherstellung verwenden
GxP-konforme elektronische Logbuch-Lösungen – von Reinraum-Logbuch-Software und QC-Labor-Logbuch-Software bis hin zu Lager-Logbuch-Software – maßgeschneidert für regulierte Fertigungsumgebungen
Arzneimittel
Durchgängiges elektronisches Logbuch für die pharmazeutische Produktion – digitalisieren Sie Forschung und Entwicklung, API, Formulierung und fertige Dosisproduktion mit GMP-konformen Funktionen des elektronischen Logbuchsystems.
Biotechnologie
Spezialisierte papierlose Logbücher in der pharmazeutischen Herstellung für Bioverarbeitungs-, Fermentations-, Zellkultur- und Biologika-Produktionsumgebungen.
Medizinische Geräte
Konforme Reinraum-Logbuchsoftware für die Herstellung, Sterilisation, Montage und Verpackung medizinischer Geräte.
Lebensmittel und Getränke
HACCP-konforme digitale Logbücher für Produktions-, Hygiene-, CIP/COP- und Umweltüberwachungs-Logbuch-Workflows in der Lebensmittelherstellung.
Gentherapie
Präzise Protokollierung für die Produktion viraler Vektoren, die Herstellung von Zelltherapien und das Kühlkettenmanagement mit elektronischem Gerätelogbuch zur Pharmakonformität.
Kosmetik & Schönheit
GMP-konforme elektronische Logbücher für die Chargenfertigung, Qualitätskontrolle und Hygieneüberwachung, unterstützt durch Logbuch-Software für die Pharmaproduktion.
"Die Compliance- und Audit-Trail-Funktionen sind hervorragend. Es unterstützt FDA 21 CFR Part 11 und EU Annex 11 und gibt uns volles Vertrauen in unsere elektronischen Aufzeichnungen."
Verifizierter Benutzer
Qualitätssicherung, Pharma
"Die abteilungsübergreifende Harmonisierung der Protokolle erfolgt nahtlos. Die Nachverfolgung der Gerätenutzung und die Integration mit unserem eDMS und LMS sorgten für eine reibungslose Einführung."
IT-Manager
Mittelständisches Pharmaunternehmen
"Die Implementierung verlief schnell und das Support-Team ist sehr reaktionsschnell. Die webbasierte Benutzeroberfläche erleichtert unseren Bedienern die täglichen Aufgaben im Logbuch."
QC-Supervisor
Unternehmenspharmazeutik
Erreichen Sie 100 % Audit-Bereitschaft mit papierlosen Logbüchern
Schließen Sie sich den führenden Life-Science-Unternehmen an, die das elektronische Logbuch AmpleLogic für die pharmazeutische Herstellung nutzen, um manuelle Compliance-Risiken zu eliminieren und die digitale Transformation mit KI-gestützten papierlosen Logbüchern in der pharmazeutischen Produktion zu beschleunigen.
Bleiben Sie in den Biowissenschaften an der Spitze
Erhalten Sie die neuesten Produktaktualisierungen, Compliance-News und Brancheneinblicke in Ihren Posteingang.