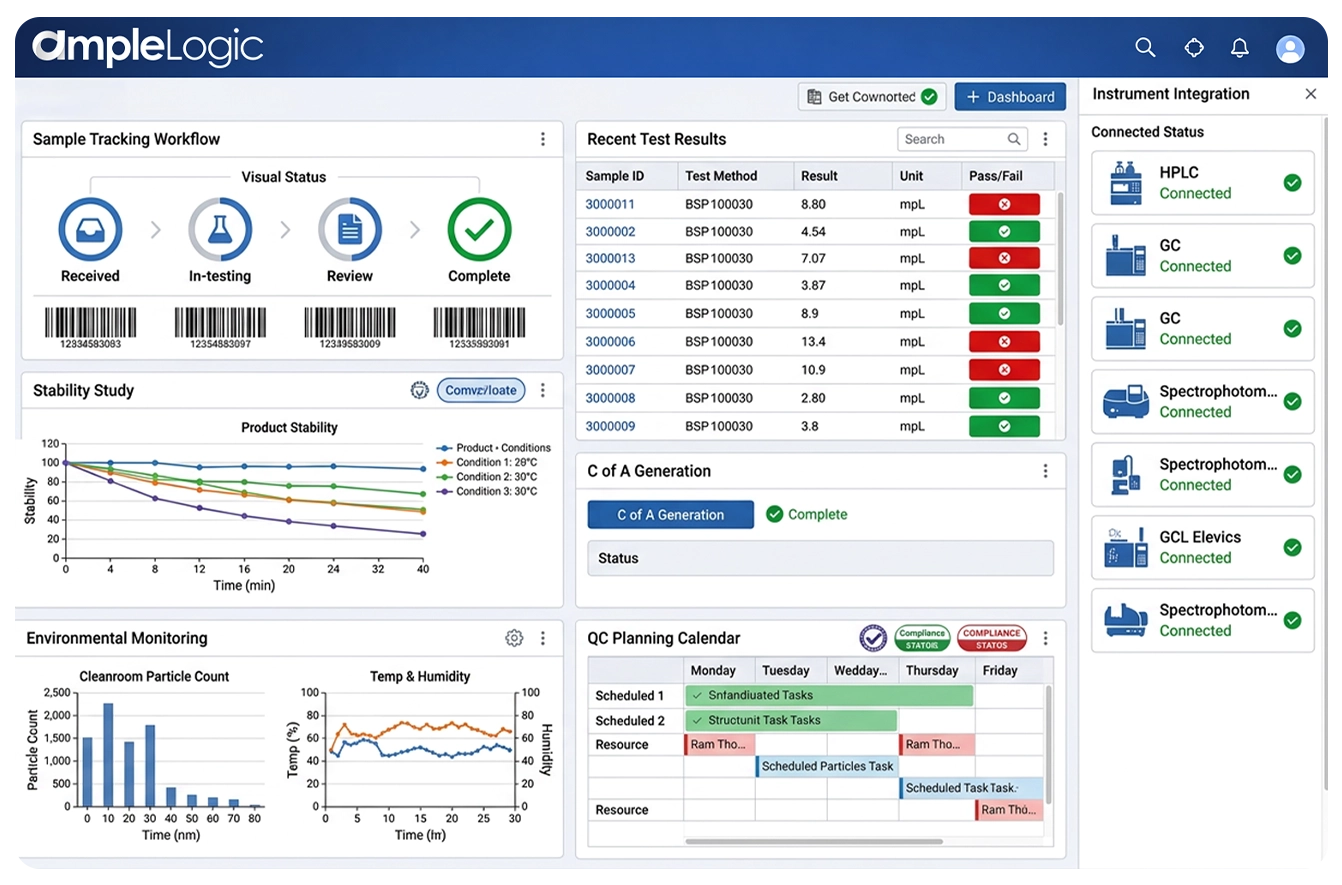
Environmental Monitoring System (EMS)-Software für die pharmazeutische Sterilherstellung
Bewahren Sie die Einhaltung von Anhang 1 und die Integrität der sterilen Fertigung mit Echtzeit-Umgebungsüberwachung, Alarmmanagement und integrierter Validierung
35 %
Weniger Chargenfehler
24/7
Kontinuierliche Überwachung
100 %
Anhang 1 Ausrichtung
90 %
Schnellere Reaktion auf Vorfälle
End-to-End Process Flow
How It Works
Structured workflow from initiation through closure with built-in compliance checkpoints and full traceability at every stage.
Installation des Sensornetzwerks
Setzen Sie Temperatur-, Feuchtigkeits-, Partikel- und Differenzdrucksensoren in allen Reinraumzonen ein
Konfiguration des Alarmschwellenwerts
Definieren Sie obere und untere Alarm-/Aktionsgrenzen für jeden Parameter entsprechend den Erwartungen in Anhang 1
Echtzeitüberwachung
Kontinuierliche Datenerfassung mit sofortiger Benachrichtigung bei Abweichungen und Parameterverstößen
Untersuchung des Vorfalls
Automatisierter Untersuchungsworkflow mit Ursachenanalyse, Folgenabschätzung und Nachverfolgung von Korrekturmaßnahmen
Verknüpfung steriler Operationen
Automatisches Halten/Freigeben von Chargenproben basierend auf dem Umgebungsüberwachungsstatus während der Herstellung
Erstellung von Regulierungsberichten
Erstellen Sie revisionssichere Umweltüberwachungsberichte und Trendanalysen für behördliche Einreichungen
Capabilities
Key Features
Comprehensive capabilities designed for regulated laboratory environments and operational excellence.
Reinraum-Umgebungsüberwachung für GMP-Pharmalabore
Echtzeitüberwachung von Umgebungsparametern mit Alarmgrenzen
Auditfähige Umweltberichte der US-amerikanischen FDA und GMP
Umfassende Dokumentation für behördliche Kontrollen
Kontaminationskontrollstrategie (CCS) für Pharmazeutik und Biowissenschaften
Entscheidende Unterstützung für die Aufrechterhaltung der Integrität einer sterilen Umgebung
Einhaltung von 21 CFR Part 11 und EU Annex 11 für die aseptische Herstellung
Angleichung an die Anforderungen des EU-Arzneimittelanhangs für die aseptische Herstellung
EMS-zu-LIMS-Integration für sterile QC-Laboroperationen
Nahtloser Datenfluss zwischen Umgebungsüberwachung und Probentests
Umweltwarnungen in Echtzeit zur Einhaltung gesetzlicher Vorschriften
Sofortige Benachrichtigung bei Abweichungen und Parameterverstößen
GMP-Umweltdatentrends und -analyse für Biowissenschaften
Langfristige Umweltdatenanalyse zur Mustererkennung
Real-World Applications
Use Cases
Proven scenarios where this module delivers measurable value across laboratory and manufacturing operations.
Injektionsherstellung – parenteral
Überwachen Sie aseptische Verarbeitungsbereiche der ISO-Klasse 5 mit kontinuierlicher Umgebungsüberwachung und chargenspezifischer Verknüpfung
Reduzieren Sie Chargenausfälle um 35 % und weisen Sie bei FDA-Inspektionen die Einhaltung nach
Sterile Compoundierung – Krankenhausapotheke
Verfolgen Sie USP <797>-verschärfende Umgebungsbedingungen mit Echtzeitwarnungen und automatisierter Dokumentation
Gewährleisten Sie die Patientensicherheit, behalten Sie die Akkreditierung bei und reduzieren Sie Fehler bei der Zusammensetzung
Herstellung viraler Vektoren – Gentherapie
Verwalten Sie komplexe Reinraumumgebungen mit mehreren Zonen mit automatisierten Umweltstatusberichten
Unterstützen Sie die Herstellung fortschrittlicher Therapien mit umfassenden Umweltnachweisen
Auftragsfertigung – CMO Operations
Standardisieren Sie die Umweltüberwachung über mehrere Kundenkampagnen hinweg mit zentralisierten Dashboards
Verbessern Sie das Kundenvertrauen, ermöglichen Sie eine schnellere Chargenfreigabe und reduzieren Sie Audit-Ergebnisse
Common Questions
Frequently Asked Questions
Ready to Modernize Environmental Monitoring System (EMS)-Software für die pharmazeutische Sterilherstellung?
See how AmpleLogic LIMS can transform your environmental monitoring system (ems)-software für die pharmazeutische sterilherstellung workflows with a personalized demo.
Bleiben Sie in den Biowissenschaften an der Spitze
Erhalten Sie die neuesten Produktaktualisierungen, Compliance-News und Brancheneinblicke in Ihren Posteingang.