Assurez-vous que chaque employé est formé et conforme GxP
Logiciel de gestion de la formation en sciences de la vie avec des capacités de logiciel de formation FDA cGMP, des attributions de formation automatisées, un suivi de la formation GMP et une gestion de la formation prête pour l'audit — un LMS conforme 21 CFR Part 11 conçu comme un LMS cloud pour les industries réglementées.
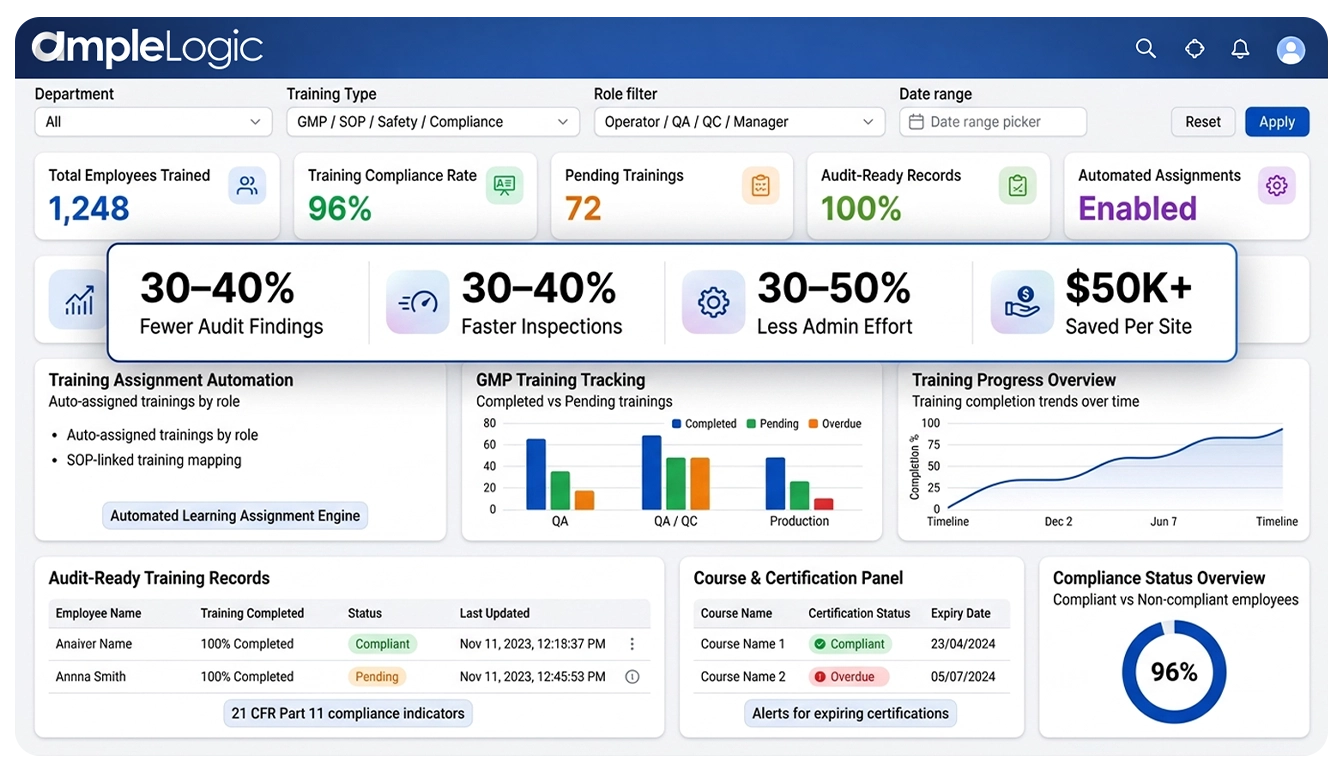
30-40%
Moins de constats d'audit
30-40%
Inspections plus rapides
30-50%
Moins d'effort administratif
$50K+
Économisé par site
État actuel de la gestion de la formation GMP dans l'industrie pharmaceutique
Les systèmes manuels et les plateformes LMS héritées créent des lacunes de conformité, des risques lors des inspections et une inefficacité opérationnelle
Manuel
La gestion de la formation des employés est suivie par des feuilles de calcul et des dossiers papier ; fragmentée, cloisonnée et à haut risque lors des inspections FDA et MHRA.
LMS hérité / partiellement intégré
LMS d'industrie réglementée partiellement automatisé avec des intégrations limitées, des lacunes entre sites et une réconciliation manuelle des dossiers de suivi de la formation GMP.
La conformité manuelle coûte plus de 50 000 $ par site
Les systèmes fragmentés sont la principale cause d'exposition aux audits et de goulots d'étranglement opérationnels dans l'industrie pharmaceutique mondiale.
La préparation manuelle aux inspections représente plus de 200 heures de travail par événement.
Plus de 50 000 $ de coûts de remédiation évités par site en éliminant les erreurs de réconciliation sur support papier.
Les silos de données fragmentés entraînent un risque d'inspection élevé en raison de dossiers inter-sites incohérents.
Défis d'intégration du LMS pharmaceutique : comment les systèmes cloisonnés créent des risques de conformité
Les données de formation, de qualité et de fabrication restent isolées dans les paysages informatiques pharmaceutiques — créant un risque systémique de conformité
Sources entrantes
ERP/RH
Doublons d'enregistrements et de rôles
LMS
Données de formation isolées
eQMS
Mises à jour de SOP non liées
RIMS
Besoins de formation réglementaire
Systèmes déconnectés
Transferts manuels entre plateformes
Intégration retardée
Les nouvelles recrues attendent des jours à des semaines
Charge d'audit lourde
Plus de 200 heures de préparation aux inspections
Risque de conformité
Lacunes de formation découvertes après exécution
Application sortante
LIMS
Qualification des analystes non vérifiée
MES
Lacunes dans l'exécution des lots
CAPS
Formation à l'étalonnage non appliquée
eLogbook
Aucune certification équipement vérifiée
Validation du nettoyage
Lacunes dans les procédures de nettoyage
Architecture de formation SOP unifiée pour l'industrie pharmaceutique
Conformité centralisée, rapports automatisés et visibilité en temps réel des compétences — dans une seule plateforme validée
Gestion centralisée de l'apprentissage
Référentiel unique pour le contenu de formation, la livraison et les capacités du logiciel de suivi de la formation dans toutes les opérations mondiales — la base d'un système de gestion de la formation qualité pour l'industrie pharmaceutique.
Contrôle d'accès basé sur les rôles
Autorisations granulaires permettant au personnel d'accéder uniquement aux formations et informations requises ; prend en charge les exigences du LMS conforme 21 CFR Part 11 et la gouvernance du système de qualification des employés pharmaceutiques.
Moteur de formation à la conformité GMP
Logiciel de formation à la conformité GMP avec évaluations automatisées des compétences, cartographie des exigences réglementaires et génération de pistes d'audit pour la préparation du système de formation à la conformité FDA.
Tableau de bord de reporting en temps réel
Visibilité instantanée sur les taux d'achèvement, le statut des compétences et les indicateurs de formation à la conformité qualité sur tous les sites — alimentant les capacités de la plateforme de formation à la préparation aux inspections.
Présence biométrique
Connectivité transparente avec les dispositifs biométriques pour un suivi de présence par authentification par empreinte digitale inviolable dans les environnements de gestion de la formation en fabrication.
Intégrations d'entreprise
Intégrations avec DMS, eQMS, ERP, RH et LIMS pour une traçabilité qualité de bout en bout dans cet écosystème de logiciel de formation à la conformité pharmaceutique.
Gestion centralisée de l'apprentissage
Référentiel unique pour le contenu de formation, la livraison et les capacités du logiciel de suivi de la formation dans toutes les opérations mondiales — la base d'un système de gestion de la formation qualité pour l'industrie pharmaceutique.
Contrôle d'accès basé sur les rôles
Autorisations granulaires permettant au personnel d'accéder uniquement aux formations et informations requises ; prend en charge les exigences du LMS conforme 21 CFR Part 11 et la gouvernance du système de qualification des employés pharmaceutiques.
Moteur de formation à la conformité GMP
Logiciel de formation à la conformité GMP avec évaluations automatisées des compétences, cartographie des exigences réglementaires et génération de pistes d'audit pour la préparation du système de formation à la conformité FDA.
Tableau de bord de reporting en temps réel
Visibilité instantanée sur les taux d'achèvement, le statut des compétences et les indicateurs de formation à la conformité qualité sur tous les sites — alimentant les capacités de la plateforme de formation à la préparation aux inspections.
Présence biométrique
Connectivité transparente avec les dispositifs biométriques pour un suivi de présence par authentification par empreinte digitale inviolable dans les environnements de gestion de la formation en fabrication.
Intégrations d'entreprise
Intégrations avec DMS, eQMS, ERP, RH et LIMS pour une traçabilité qualité de bout en bout dans cet écosystème de logiciel de formation à la conformité pharmaceutique.
Capacités des modules LMS AmpleLogic
Gestion complète du cycle de vie de la formation, de l'espace de travail des employés aux tableaux de bord exécutifs
Mes besoins en formation
Votre espace de travail de formation personnalisé
Tableau de bord personnalisé affichant les cours attribués, les échéances à venir, les certifications pharmaceutiques des employés complétées et le statut des compétences en un coup d'œil dans ce logiciel de gestion de la formation pour les sciences de la vie.
Données de référence
Configuration organisationnelle centralisée
Détails des employés
Système de qualification des employés pharmaceutiques
Manuel de formation
Référentiel de gestion de la formation SOP
Sessions de formation
Moteur d'exécution de formation multimode
Certifications
Certifications des employés pharmaceutiques et validation de la conformité
Indicateurs
Suivi de la formation GMP et surveillance des performances
Analytique
Analyses du système de gestion de l'apprentissage IA
Tableaux de bord interactifs
Visibilité en temps réel de la formation et de la conformité
Expérience d'apprentissage GMP moderne et flexible
Conçu pour améliorer l'engagement, la préparation aux compétences et l'efficacité de la formation selon les rôles avec la création de contenu alimentée par l'IA
Formats d'apprentissage multiples
Prend en charge l'apprentissage basé sur des documents, les formations enregistrées, les podcasts générés par l'IA (multilingue) et la formation en situation de travail (OJT) — un LMS complet pour les industries réglementées offrant une flexibilité maximale.
Génération de questionnaires basée sur l'IA
Système de gestion de l'apprentissage IA avec création automatisée de banques de questions à partir du contenu de formation, réduisant les efforts manuels jusqu'à 80 % tout en maintenant la qualité des évaluations et l'alignement réglementaire.
Prise en charge de l'apprentissage basée sur des standards
Conformité totale SCORM et xAPI pour une interopérabilité de contenu sans accroc dans ce logiciel de formation à la qualité pharmaceutique avec suivi analytique aux standards de l'industrie.
Parcours d'apprentissage personnalisés
Parcours d'apprentissage basés sur le rôle, le département et le stade de carrière qui s'adaptent aux lacunes de compétences individuelles — offrant une gestion de la formation des employés qui améliore la pertinence et les taux d'achèvement des cours.
Podcasts générés par l'IA
Convertissez automatiquement les supports de formation en contenu audio multilingue engageant, permettant un apprentissage en déplacement pour le personnel de terrain et de gestion de la formation en fabrication.
Évaluations interactives
Quiz configurables, évaluations basées sur des scénarios et démonstrations pratiques avec notation automatisée et flux de travail de validation des compétences pour la conformité du système de qualification des employés pharmaceutiques.
Intelligence de formation en temps réel et suivi des performances
Visibilité en temps réel sur les performances de la formation, le statut de conformité et la préparation de la main-d'œuvre
Rapports de conformité et d'audit
Générez instantanément des preuves prêtes pour les régulateurs avec des dossiers complets d'achèvement de formation, une documentation des compétences et des résumés de pistes d'audit.
Rapports de performance et de qualification des employés
Assurez-vous que chaque employé est formé et qualifié pour les rôles assignés grâce à des analyses de performance détaillées et une analyse des lacunes de qualification.
Suivi des qualifications et certifications
Maintenez des dossiers de certification vérifiés sur tous les sites avec des notifications automatiques d'expiration et une planification de la requalification.
Planification des cursus et opérations de formation
Optimisez les programmes de formation et la planification des ressources avec des prévisions de la demande, l'utilisation des formateurs et les indicateurs d'efficacité des cours.
Construction de systèmes de formation prêts pour l'audit
Concevez une formation pour la préparation aux audits FDA 21 CFR Part 11 et GMP avec génération automatique de preuves
Génération automatisée de pistes d'audit
LMS conforme 21 CFR Part 11
Le LMS conforme 21 CFR Part 11 enregistre les formations, les évaluations et les dossiers de compétences avec des horodatages et des identifiants utilisateurs pour une traçabilité complète en tant que logiciel de formation FDA cGMP.
Reporting de conformité en temps réel
Logiciel de formation à la conformité GMP
Le tableau de bord du logiciel de formation à la conformité GMP indique qui est formé, le statut des compétences, les expirations et les éléments en retard sur tous les sites pour une gestion de la formation prête pour l'audit en temps réel.
Exigences GMP intégrées
Logiciel de formation FDA cGMP
Modules GMP préconfigurés et critères d'évaluation alignés en tant que logiciel de formation à la qualité pharmaceutique — réduisant les efforts de validation et accélérant le déploiement avec la prise en charge de la formation de recyclage GMP.
Contrôle de version du contenu de formation
Mises à jour de la gestion de la formation SOP
Historique complet des modifications de contenu avec dates d'entrée en vigueur, approbations et déclencheurs automatiques de reformation lors des révisions de SOP — essentiel à la gestion de la formation SOP et à la formation à la conformité qualité.
Contrôles d'accès basés sur les rôles
Intégrité des données et logiciel de formation GxP
Logiciel de formation GxP avec des autorisations granulaires garantissant que seul le personnel autorisé accède aux données de formation sensibles, soutenant l'intégrité des données ALCOA+ dans ce système de formation à la conformité FDA.
Plateforme de formation à la préparation aux inspections
Inspections plus rapides et moins de constats
La plateforme de formation à la préparation aux inspections permet de répondre aux questions des inspecteurs en quelques minutes ; le temps d'audit réduit de 30 à 40 % ; remédiation plus rapide et présentation des preuves pour une gestion de la formation prête pour l'audit.
Analyses basées sur l'IA pour la gestion prédictive des compétences
Passez d'une formation réactive à des interventions opportunes axées sur la conformité grâce à l'intelligence IA intégrée
Capacités prédictives
- Le système de gestion de l'apprentissage IA identifie les lacunes de compétences émergentes avant qu'elles n'impactent les activités de laboratoire, de production ou de maintenance
- Prévision de la demande de formation basée sur les changements de main-d'œuvre, les mises à jour réglementaires et les exigences du calendrier annuel de formation GMP
Signaux de risque et de conformité
- Détecte automatiquement les schémas inhabituels de formation et de qualification dans ce logiciel de formation GxP
- Signale les prérequis manquants, la formation de recyclage GMP en retard ou les séquences de formation non conformes
- Surveille les mises à jour réglementaires et recommande des ajustements de formation pour l'alignement du système de formation à la conformité FDA
Indicateurs opérationnels et de tableau de bord
- Logiciel de suivi de la formation avec tendances d'efficacité et de délai d'acquisition des compétences
- Logiciel de matrice de formation avec cartes thermiques des compétences par site et par rôle
- Indicateurs de la plateforme de formation à la préparation aux inspections et tendances des retours d'audit

Impact métier et ROI avec le système de gestion de l'apprentissage GMP AmpleLogic
Conformité tangible, efficacité et protection des coûts pour les CXO et les responsables AQ utilisant le meilleur LMS pour les entreprises pharmaceutiques
30-40%
Réduction des constats d'audit liés à la formation
Baisse significative des constats de conformité grâce aux attributions de formation automatisées, à la génération de preuves et à la visibilité en temps réel des compétences dans ce système de gestion de la formation qualité pour l'industrie pharmaceutique.
30-40%
Temps de réponse aux inspections plus rapides
Livraison plus rapide des preuves aux inspecteurs avec un accès instantané aux dossiers de suivi de la formation GMP, aux pistes d'audit et à la documentation de qualification via la gestion de la formation prête pour l'audit.
30-50%
Réduction de l'effort d'administration de la formation
Charge de travail réduite pour les équipes de formation grâce aux attributions de formation automatisées, à la planification, au suivi des compétences et aux flux de travail de reporting du logiciel de suivi de la formation.
$50K+
Coût de remédiation évité par site
Protection des coûts grâce à la remédiation évitée en éliminant les erreurs de réconciliation sur support papier et les lacunes de formation manuelle avec ce LMS cloud pour les industries réglementées.
4-6 sem.
Intégration plus rapide et qualité améliorée
Bénéfices opérationnels sur tous les sites et système cohérent de qualification des employés pharmaceutiques dans les laboratoires, la production et les équipes de gestion de la formation en fabrication.
10 mois
Calendrier de retour sur investissement complet
Passez du fragmenté à l'intégré : une seule plateforme unifiée de logiciel de gestion de la formation en sciences de la vie avec une mise en œuvre en 4 à 6 semaines et un retour sur investissement complet en 10 mois.
aPaaS pour les sciences de la vie — LMS cloud pour les industries réglementées
Application de la qualification de bout en bout dans tout l'écosystème d'entreprise avec un logiciel de formation à la conformité pharmaceutique
DMS
inboundBesoins de formation issus de l'introduction de nouvelles SOP et des changements de version — reformation automatiquement déclenchée lors des mises à jour de documents.
eQMS
inboundBesoins de formation issus du contrôle des modifications, des constats d'audit, des déviations et des événements OOS/OOT pour des actions correctives.
RIMS
inboundBesoin de formation issu des constats réglementaires — cartographie automatique des exigences réglementaires aux cursus de formation.
LIMS
outboundValidation en temps réel de la qualification des analystes — garantit que seuls des analystes formés exécutent les procédures de laboratoire.
eLog Book
outboundContrôle d'accès aux équipements — vérifie la certification de formation avant d'accorder l'accès à l'enregistrement de l'équipement.
Validation du nettoyage
outboundApplication du personnel formé pour les procédures de nettoyage — bloque les opérateurs non formés de l'exécution des protocoles.
eBMR / MES
outboundApplication de la certification des opérateurs — valide les qualifications du personnel de fabrication avant l'exécution des lots.
CAPS
outboundApplication de la formation à l'étalonnage — garantit que les techniciens sont formés avant d'effectuer des activités d'étalonnage.
Indicateurs qualité
outboundLes données d'efficacité de la formation alimentent les tableaux de bord qualité — corrèle l'achèvement de la formation avec les KPI qualité.
SAP / ERP
BidirectionnelSynchronisation bidirectionnelle des données des employés, alignement des centres de coûts et gestion de la hiérarchie organisationnelle.
Systèmes RH
BidirectionnelLes déclencheurs d'intégration des employés, les changements de rôle et les flux de travail de fin de contrat mettent automatiquement à jour les attributions de formation.
UMS
BidirectionnelGestion unifiée des utilisateurs avec authentification unique, provisionnement des accès et gouvernance des identités dans tous les modules.
Principaux différenciateurs fournisseurs
Ce qui distingue le logiciel de formation à la qualité pharmaceutique AmpleLogic des plateformes de gestion de l'apprentissage traditionnelles et héritées
Intégration aPaaS la plus approfondie
Contrairement aux concurrents limités à l'intégration QMS uniquement, AmpleLogic connecte ce système de gestion de l'apprentissage GMP nativement à plus de 14 modules incluant LIMS, eBMR/MES, CAPS et la validation du nettoyage — offrant une application de la qualification de bout en bout.
Intelligence native IA
Agents de système de gestion de l'apprentissage IA intégrés pour la génération de questionnaires, la création de podcasts multilingues, la gestion prédictive des compétences et l'analyse automatisée des lacunes de formation — allant au-delà de l'automatisation de base dans les logiciels de formation à la conformité pharmaceutique.
Application de la qualification
Blocage en temps réel du personnel non qualifié dans la gestion de la formation en fabrication (MES), la gestion de la formation en laboratoire (LIMS), l'étalonnage (CAPS) et les opérations de nettoyage — une capacité que les concurrents n'offrent pas au point d'exécution.
Délai rapide de création de valeur
Mise en œuvre en 4 à 6 semaines avec des modules GMP préconfigurés, des modèles validés et une documentation alignée GAMP 5 — contre des délais de 6 à 12 mois typiques des fournisseurs de LMS d'industries réglementées héritées.
Les industries que nous servons
Solutions de formation conformes GxP dans les sciences de la vie et les industries réglementées
Pharmaceutique
Meilleur LMS pour les entreprises pharmaceutiques avec une formation conforme GMP pour les équipes de fabrication et de qualité
Biotechnologie
Logiciel de formation GxP avec des parcours de qualification complexes pour la recherche et la production
Dispositifs médicaux
Formation alignée ISO 13485 et gestion de la formation des employés pour les entreprises de dispositifs
CRO / CDMO
Ségrégation de la formation multi-client avec des dossiers de gestion de la formation prêts pour l'audit
Sciences de la vie
Logiciel de gestion de la formation pour les sciences de la vie avec gouvernance de l'apprentissage à l'échelle de l'entreprise sur les sites mondiaux
Fabrication
Gestion de la formation en fabrication avec certification des opérateurs et suivi de la qualification des équipements
"Tableaux de bord conviviaux et interface intuitive pour la gestion de la conformité en matière de formation. Les capacités de reporting sont excellentes pour la préparation aux audits."
Responsable formation
Pharma marché intermédiaire
"Une mise en œuvre rapide et un support réactif 24h/24 7j/7 ont rendu la transition sans accroc. La conformité aux normes réglementaires pharmaceutiques sans paramétrage supplémentaire est un avantage considérable."
Responsable assurance qualité
Sciences de la vie
"L'intégration avec d'autres modules AmpleLogic comme eQMS et DMS nous fait gagner un temps considérable. L'attribution et le suivi automatisés de la formation sont excellents."
Directeur L&D
Entreprise pharmaceutique
Favorisez des décisions plus intelligentes grâce à l'intelligence de formation en temps réel
Rejoignez les grandes organisations des sciences de la vie qui utilisent AmpleLogic — le meilleur LMS pour les entreprises pharmaceutiques — pour accélérer la formation à la conformité qualité, réduire le risque d'audit et construire une culture axée sur les compétences avec un logiciel de gestion de la formation pour les sciences de la vie.
Gardez une longueur d'avance dans les sciences de la vie
Recevez les dernières mises à jour produit, actualités de conformité et analyses du secteur directement dans votre boîte mail.