IA d'Entreprise Alimentée par Solutions pour Chaque Flux de Travail Pharmaceutique
Suite complète de solutions logicielles validées pour les organisations pharmaceutiques, biotechnologiques et des sciences de la vie.
aPaaS Low Code pour les Sciences de la Vie — Architecture d'Intégration Unifiée
Les modules d'AmpleLogic ne fonctionnent pas en silos. Chaque solution est interconnectée — partageant des données, déclenchant des flux de travail et maintenant une source unique de vérité à travers les fonctions Qualité, Fabrication et Laboratoire QC.
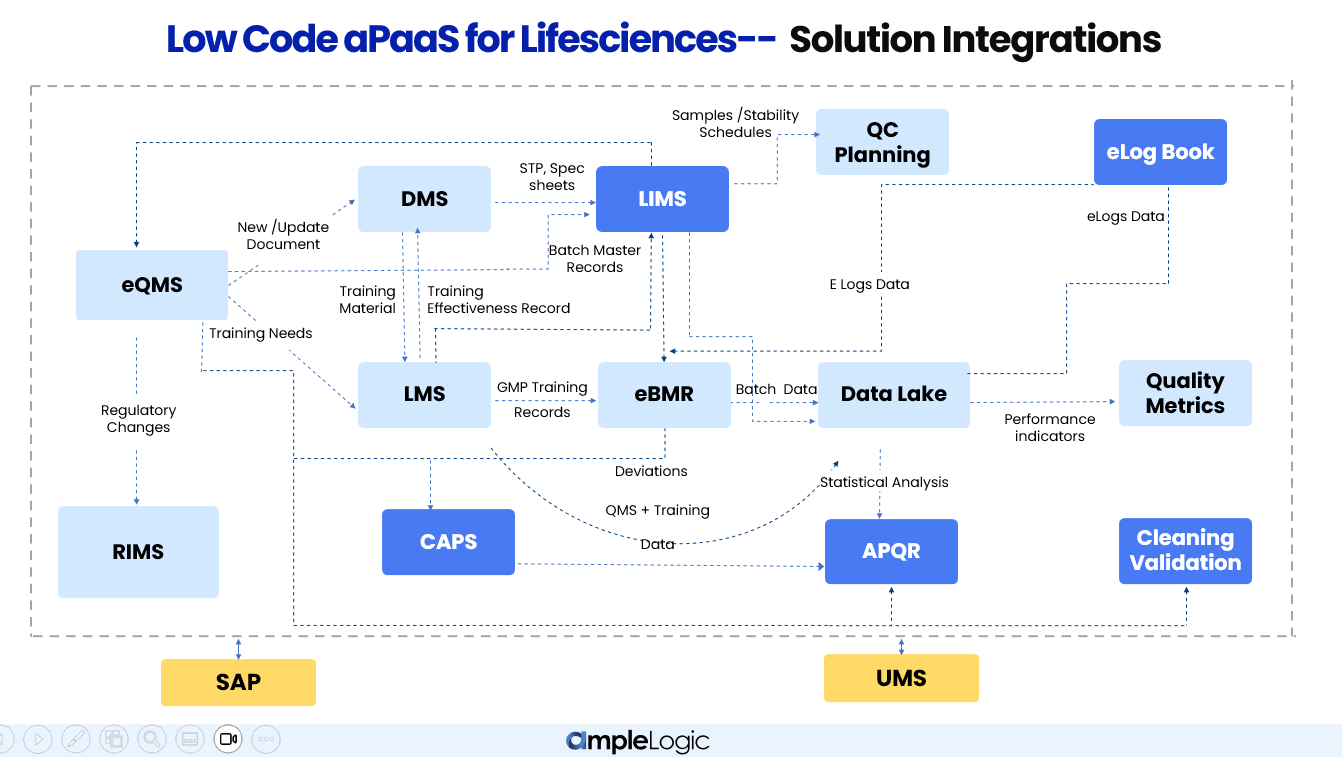
Comment les modules AmpleLogic s'intègrent à travers les fonctions Qualité, Fabrication et Laboratoire QC dans les entreprises pharmaceutiques
Lac de Données Unifié
Tous les modules alimentent un lac de données centralisé, permettant des indicateurs de performance en temps réel, des métriques qualité et des analyses transversales sans consolidation manuelle des données.
Cascades de Flux de Travail Automatisés
Un seul événement — tel qu'une déviation en fabrication — déclenche automatiquement le CAPA dans eQMS, la révision de document dans DMS, la reformation dans LMS et l'évaluation d'impact dans APQR.
Flux de Données Bidirectionnel
Les données circulent dans les deux sens entre les modules. Par exemple, LMS renvoie les enregistrements d'efficacité de formation à eQMS, et LIMS retourne les résultats analytiques à eBMR pour la libération des lots.
Comment les Intégrations Fonctionnent à Travers les Fonctions
Gestion de la Qualité
L'écosystème de gestion de la qualité garantit que chaque déviation, CAPA, contrôle des changements et constatation d'audit est suivi de bout en bout. eQMS est au centre — déclenchant les flux documentaires dans DMS, les affectations de formation dans LMS, et alimentant les données statistiques dans APQR pour les revues annuelles de qualité produit.
Document Nouveau / Mis à Jour
Lorsqu'une déviation ou un contrôle des changements est soulevé dans eQMS, DMS initie automatiquement un flux de révision SOP, garantissant que les documents restent à jour avec chaque événement qualité.
Besoins en Formation
Les SOP nouveaux ou révisés déclenchent des affectations de formation automatiques dans LMS. Les enregistrements d'efficacité de formation retournent vers eQMS pour clore la boucle qualité.
Analyse Statistique
Les données de lot, les déviations, les enregistrements CAPA et les paramètres de processus sont agrégés dans le lac de données et alimentés dans APQR pour des revues annuelles complètes de qualité produit.
Données QMS + Formation
Les déviations d'étalonnage et les enregistrements de maintenance préventive issus de CAPS s'intègrent dans APQR, offrant une image complète de l'impact des équipements et instruments sur la qualité du produit.
Gardez une longueur d'avance dans les sciences de la vie
Recevez les dernières mises à jour produit, actualités de conformité et analyses du secteur directement dans votre boîte mail.