From Preventive to Predictive — Smarter Equipment Maintenance
Transform calibration and preventive maintenance into a unified asset intelligence system. AmpleLogic leverages AI, automation, and predictive analytics to monitor equipment health, optimize maintenance schedules, and ensure continuous GMP compliance across your manufacturing operations.
3-5x
Return on Investment
70%
Faster Approvals
60%
Emergency Cost Reduction
45%
Less Unplanned Downtime
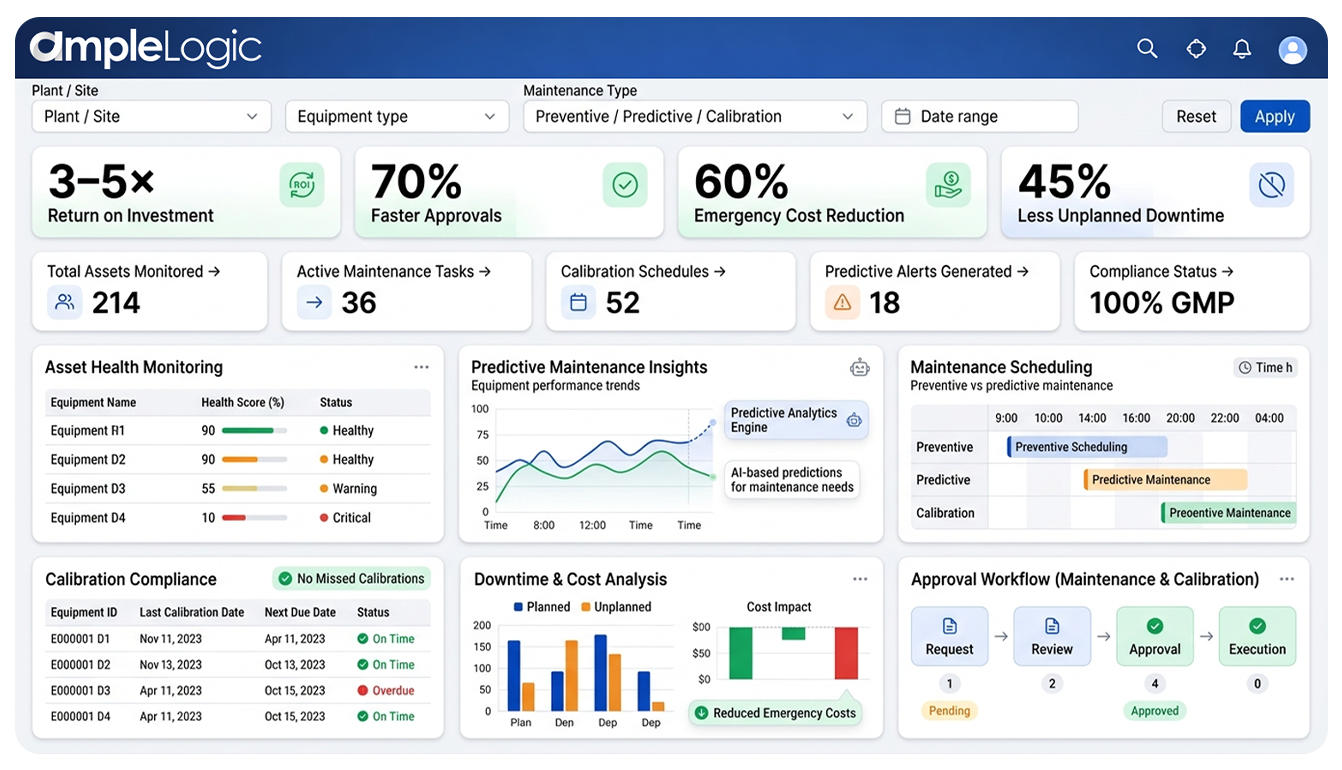
From Traditional Calibration & Preventive Maintenance to AI-Driven Asset Intelligence
Traditional maintenance systems only record calibration and maintenance activities. AmpleLogic goes further by providing an AI-driven GMP asset intelligence platform that continuously analyzes equipment data, predicts maintenance needs, and helps teams make proactive decisions to improve reliability, compliance, and operational efficiency.
By combining calibration management, preventive maintenance automation, and intelligent analytics, the platform delivers complete visibility into the health and performance of critical GMP assets across facilities.
AI-Driven GMP Asset Intelligent Maintenance Insights
Advanced analytics evaluate equipment performance trends and recommend optimal maintenance schedules before failures occur.
Predictive Maintenance Intelligence
Identify potential equipment issues early using predictive analytics, reducing unplanned downtime and ensuring consistent manufacturing performance.
Manual Calibration and Preventive Maintenance Tracking Increases GMP Audit Risks
Traditional paper-based methods are no longer sufficient for the strict requirements of the life sciences industry
Manual Paper Tracking
Manual paper tracking leads to frequent errors and massive time consumption in record keeping.
Audit Readiness Gaps
Difficulty in maintaining audit readiness due to lack of transparent and verifiable documentation.
Missed Calibrations
Inefficient scheduling causes missed calibrations, leading to unexpected equipment malfunctions.
No Real-Time Visibility
Lack of real-time visibility into performance makes it impossible to anticipate critical maintenance needs.
Calibration & Preventive Maintenance (CAPS) Framework for GMP Compliance
Purpose-built solutions offer deep functionality tailored to regulatory compliance and the specific needs of calibration standards
21 CFR Part 11
Built-in digital signatures and audit trails for seamless regulatory compliance.
WHO & EU GMP Guidelines
Calibrated, maintained, and qualified equipment; preventive maintenance schedules defined and followed.
Digital Checklists
Mobile-ready user interfaces that simplify technician data entry and reduce error.
Audit Readiness
Unified view of maintenance and quality, reducing "double downtime."
Features of AmpleLogic Calibration & Preventive Maintenance Software
Comprehensive calibration and preventive maintenance with AI-powered compliance automation
Job Order Management
Easily create, assign, and track work orders for efficient execution of maintenance tasks and streamlined workflows.
Preventive Maintenance
Maintenance Planning
Centralized Dashboards
RFID & Barcode Tracking
Integration Capabilities
How Preventive Maintenance Evolves to Predictive Maintenance in the Pharma Industry
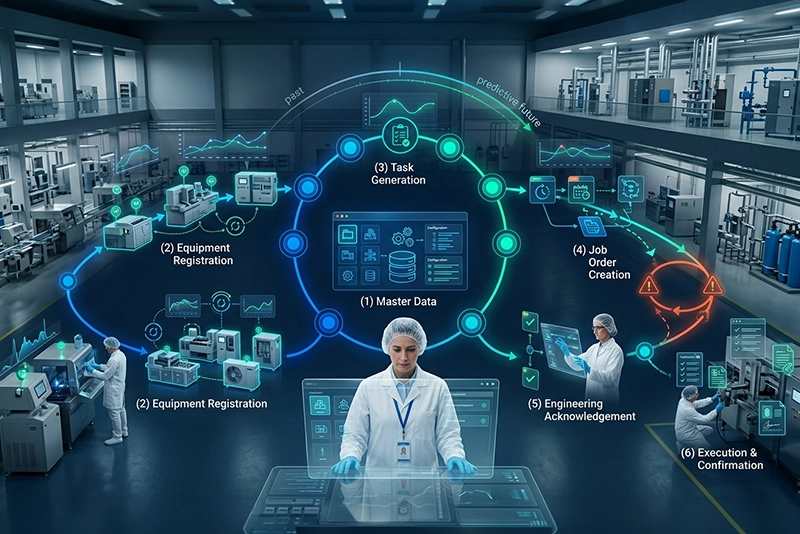
End-to-end calibration and maintenance workflow from master data to confirmation
Master Data
Define equipment, instruments, and maintenance parameters in the central registry.
Equipment / Instrument Registration
Register all assets and link equipment with frequencies and checkpoints.
Task Generation Based on Frequency
System auto-generates tasks based on configured calibration and maintenance frequencies.
Raise Job Order (Schedule / Unschedule)
Create and dispatch scheduled or unscheduled job orders with clearance workflows.
Engineering Acknowledgement
Engineers acknowledge and route to internal checkpoints or external attachments.
Execution & Confirmation
Execute checkpoints, attach documentation, and confirm completion with digital signatures.
Master Data
Define equipment, instruments, and maintenance parameters in the central registry.
Equipment / Instrument Registration
Register all assets and link equipment with frequencies and checkpoints.
Task Generation Based on Frequency
System auto-generates tasks based on configured calibration and maintenance frequencies.
Raise Job Order (Schedule / Unschedule)
Create and dispatch scheduled or unscheduled job orders with clearance workflows.
Engineering Acknowledgement
Engineers acknowledge and route to internal checkpoints or external attachments.
Execution & Confirmation
Execute checkpoints, attach documentation, and confirm completion with digital signatures.
Note: If the tolerance period for acknowledgment or execution is exceeded, the process will automatically trigger a deviation.
Business and Compliance Benefits of AI-Driven Calibration & Preventive Maintenance Software
Compliance, rapid upgrades, deep integration support
Ensures GMP & Regulatory Compliance
Built-in regulatory workflows ensure every maintenance activity aligns with GMP requirements.
Built-In Checklists & Guided Execution
Configurable checklists guide technicians through each step, eliminating errors and omissions.
Audit-Ready Documentation & Traceable Records
Complete digital records with timestamps, signatures, and full traceability for any audit.
Automated Deviation / CAPA Triggers
Automatic escalation to deviation and CAPA workflows when tolerances are exceeded.
Reduces Regulatory Risk & Observations
Proactive compliance monitoring minimizes the risk of regulatory findings and observations.
Minimizes Downtime, Improves Batch Release Speed
Preventive maintenance reduces unplanned stoppages and accelerates production cycles.
Enhances Visibility & KPIs for Management
Real-time dashboards provide management with actionable insights into equipment health and compliance.
Increases Technician Efficiency
Mobile-ready interfaces and guided workflows help technicians complete tasks faster and more accurately.
CAPS Integrated Platform: Enterprise Integrations with eQMS, LIMS, eBMR & DMS
End-to-end equipment lifecycle enforcement across the entire enterprise ecosystem
DMS
outboundAutomates archival of certificates and logs; ensures latest SOPs are always available.
LMS
outboundVerifies technician training and certification before allowing task execution.
eQMS
outboundTriggers Deviation/CAPA workflows automatically for failures and missed preventive maintenance.
LIMS
outboundBlocks instrument use if calibration status is "Expired" or "Failed" — ensuring data integrity.
eBMR / MES
outboundSynchronizes equipment status to ensure only validated equipment is used in manufacturing.
SAP / ERP
Bi-directionalBi-directional sync of equipment master data, cost centers, and maintenance history.
UMS
Bi-directionalUnified user management with single sign-on and access provisioning across maintenance modules.
ROI of AI-Driven Calibration & Maintenance Automation
Digitalizing your maintenance operations yields high returns through efficiency gains and risk mitigation
Administrative Savings
Reduce total labor costs by automating approval workflows and documentation tasks.
Risk Mitigation
Minimize the potential for massive non-compliance penalties and costly product scrap rates.
Downtime Reduction
Prevent expensive production halts by using AI to predict and prevent equipment failure.
3-5x
Return on Investment
Every dollar invested in CAPS delivers 3-5x return through reduced emergency repairs, minimized downtime, and extended equipment lifespan.
70%
Faster Approvals
Achieve 70% faster approval workflows through digital signatures, automated routing, and elimination of paper bottlenecks.
60%
Emergency Maintenance Cost Reduction
60% reduction in emergency maintenance costs through preventive scheduling and AI-powered predictive maintenance.
45%
Decrease in Unplanned Downtime
45% decrease in unplanned downtime across operations through automated scheduling and real-time equipment monitoring.
Is Your Current Preventive Maintenance System Enough for GMP Calibration & 21 CFR Part 11 Compliance?
Compare traditional maintenance systems against AmpleLogic's purpose-built calibration and PM solution
| Requirements | Standard PM System | AmpleLogic Calibration / PM |
|---|---|---|
| Formal calibration workflows | Loosely controlled | Workflow driven |
| Audit trails | Limited audit | GMP audit trail |
| Checklists and instructions | Uncontrolled steps | Configured checklists |
| Formal calibration workflows | Manual verification | Auto compliance |
| Calibration tolerance checks | Not compliant | 21 CFR compliant |
| Pass/Fail decisions & history | Status based | Decision controlled |
Success Story: Laurus Labs Achieves GMP Compliance with AmpleLogic CAPS
Laurus Labs successfully digitized their manual processes to achieve world-class GMP compliance.
The Challenge
Needed to digitize manual paper-based processes for calibration to increase efficiency and reduce errors.
The Solution
Implementation of AmpleLogic CAPS as their product from the AmpleLogic suite over 4 years.
The Result
Achieved full GMP compliance and streamlined operations through a trusted digital partnership.
Why AmpleLogic CAPS Software Is the Best Calibration & Preventive Maintenance Software
What sets AmpleLogic CAPS apart from generic maintenance and CMMS platforms
Deepest aPaaS Integration
Unlike standalone maintenance tools, AmpleLogic CAPS connects natively to DMS, LMS, eQMS, LIMS, eBMR/MES — providing end-to-end equipment lifecycle enforcement.
AI-Powered Predictive Maintenance
Built-in AI agents predict equipment failure patterns, optimize maintenance schedules, and reduce unplanned downtime through data-driven insights.
Automated Compliance Enforcement
Real-time blocking of uncalibrated equipment across LIMS, eBMR/MES, and lab operations — ensuring only validated instruments are used at the point of execution.
Rapid Time to Value
Implementation with pre-configured GMP modules, validated templates, and GAMP 5-aligned documentation — delivering value in weeks, not months.
Industries Using Calibration & Preventive Maintenance Software
GxP-compliant calibration and maintenance solutions across Life Sciences and regulated industries
Pharmaceuticals
GMP-compliant calibration and PM for manufacturing
Biotechnology
Precision instrument management for biotech labs
Medical Devices
ISO 13485-aligned equipment qualification
CRO / CDMO
Multi-client equipment segregation and audit trails
Life Sciences
Enterprise-wide maintenance governance across sites
Manufacturing
Preventive maintenance for production equipment
"User-friendly with great dashboards and support. The system-generated schedules and reminders ensure we never miss a calibration deadline."
Instrument Engineer
Mid-Market Pharmaceutical
"Direct data capturing for calibration and preventive maintenance eliminates papers entirely. The SAP and QMS integration is seamless."
QA Manager
Enterprise Life Sciences
"Easy to use interface with helpful dashboards for tracking all instruments and equipment. Implementation was smooth and the support team is excellent."
Calibration Lead
Pharmaceutical Manufacturing
Secure Your GMP Calibration & Compliance Future
Schedule a free demo and consultation today to see how AmpleLogic CAPS can transform your maintenance operations.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.