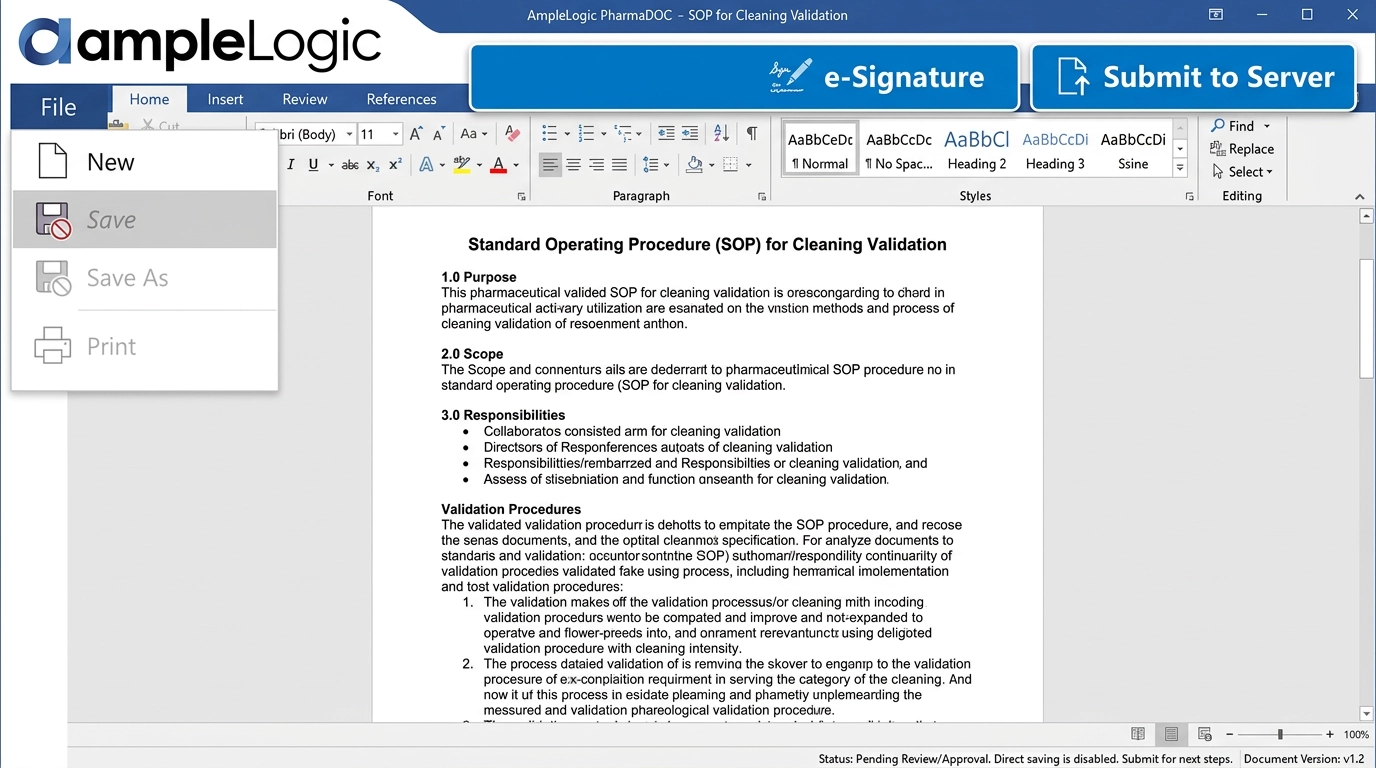
Control Every Document — From Authoring to Retirement
AmpleLogic's pharmaceutical document management software delivers enterprise-grade document control software with pharma-grade compliance, barcoding, controlled SOP management, print issuance, version and superseding controls, and intelligent automation.
25%
Audit Findings Reduction
30%
Time to Release Improvement
50%
Manual Effort Saved
FDA
21 CFR Part 11 Ready
Key GMP Challenges Without Pharmaceutical Document Control Software
Critical pain points impacting compliance, efficiency, and traceability in pharmaceutical document management
Paper-Based or Partially Digital Pharma Document Systems Still Persist
Without a compliance document management system, manual document tracking consumes 15-20 hours per week, increasing errors and retrieval time.
EDMS Feature Gaps: Weak SOP Version Control and Limited Print Issuance
Outdated SOPs used by operators due to lack of document version control software, leading to audit findings and compliance violations.
No Barcode-Based Document Tracking and Weak Superseding Management
Obsolete documents remain in circulation with no controlled copy tracking or automated retirement.
Difficult DMS Integration with LMS, eQMS, LIMS, and MES Systems
Disconnected systems break traceability, increasing audit preparation effort and response time.
What a Pharma-Grade EDMS Must Deliver for GMP Compliance
Document lifecycle management, regulatory compliance, controlled SOP management, and validated automation
Document Lifecycle Management
Complete document lifecycle management software covering authoring to review to approval to release to archive with controlled superseding and obsolescence.
Validation and Controls
Validated document approval workflow software with mapped controls to regulatory requirements and digital document control system capabilities.
Integrations and Automation
Seamless system integrations, advanced search, and AI-driven automation to reduce manual reconciliation.
21 CFR Part 11 and EU Annex 11 Regulatory Compliance
A 21 CFR Part 11 compliant document management system meeting FDA, EU Annex 11, and GxP document management system requirements with mapped controls.
Traceability and Security
Robust document version control software, audit trails, electronic signatures, and controlled copies print and retrieval tracking.
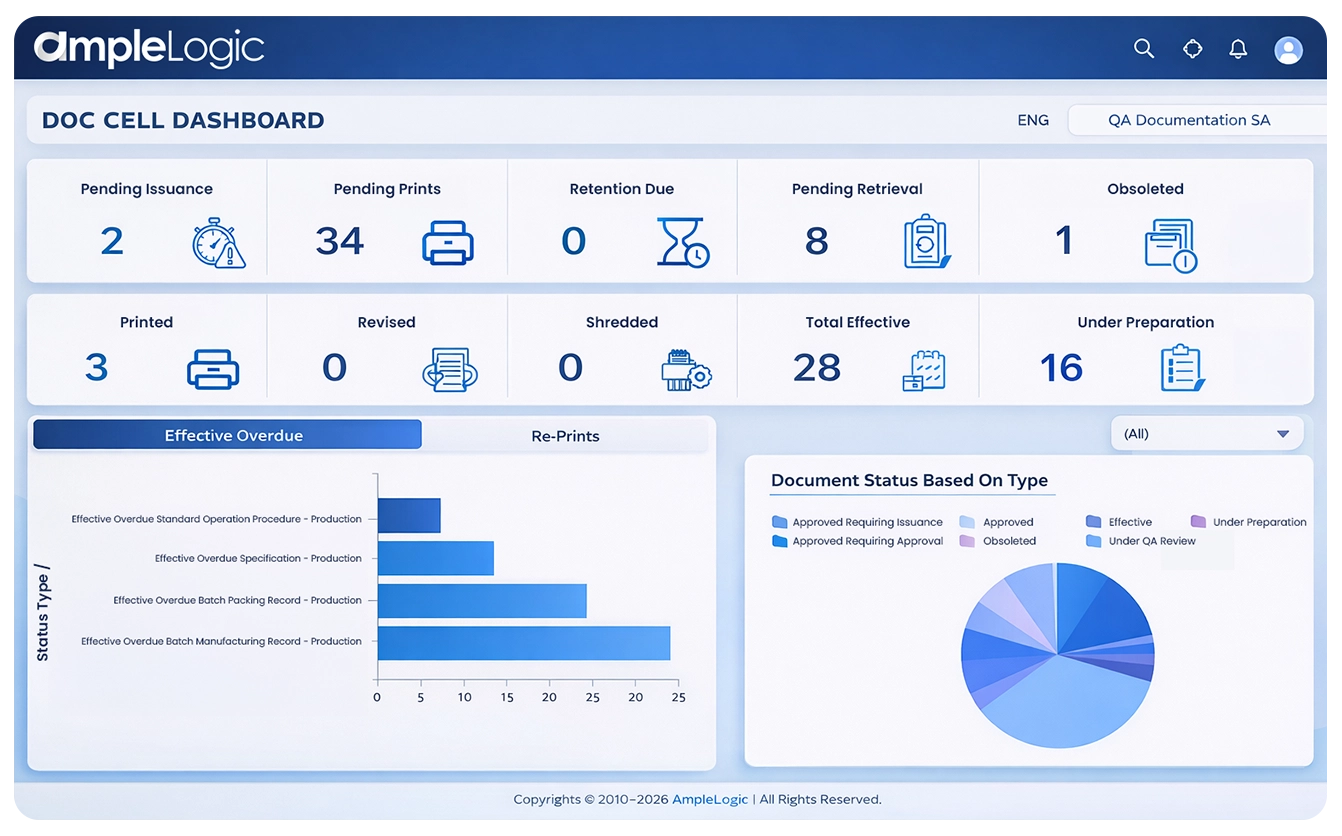
Core GMP Document Types Managed in Pharmaceutical Document Management Software
Document lifecycle management software with audit trails and metadata for GMP readiness
Quality Documents
- Standard Operating Procedures (SOPs) with version control
- Change Control records with approval workflows
- Linked training records for compliance traceability
- Obsolete copy management and superseding control
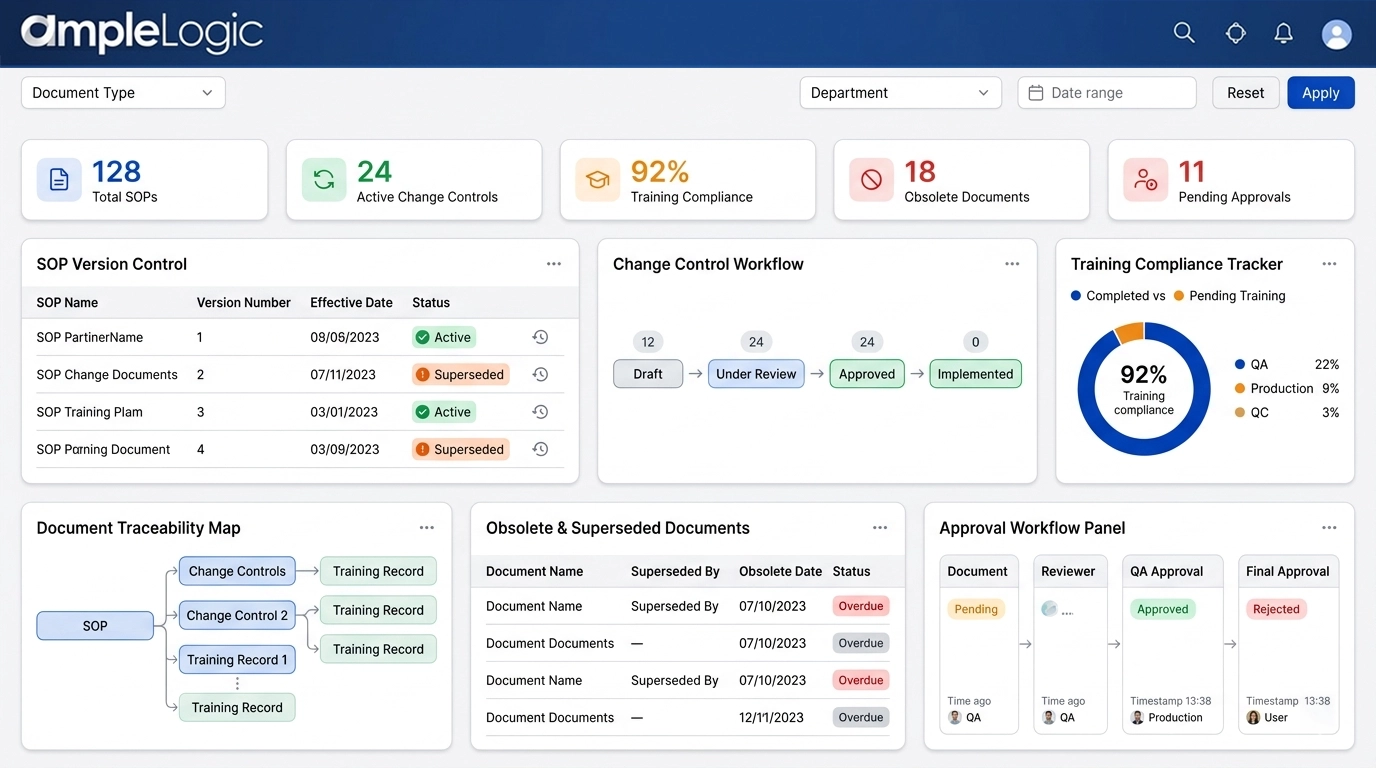
SOP Version Control Software: Superseding, Obsolete Copy, and Lifecycle Controls
Ensure only current documents drive regulated processes with digital document control
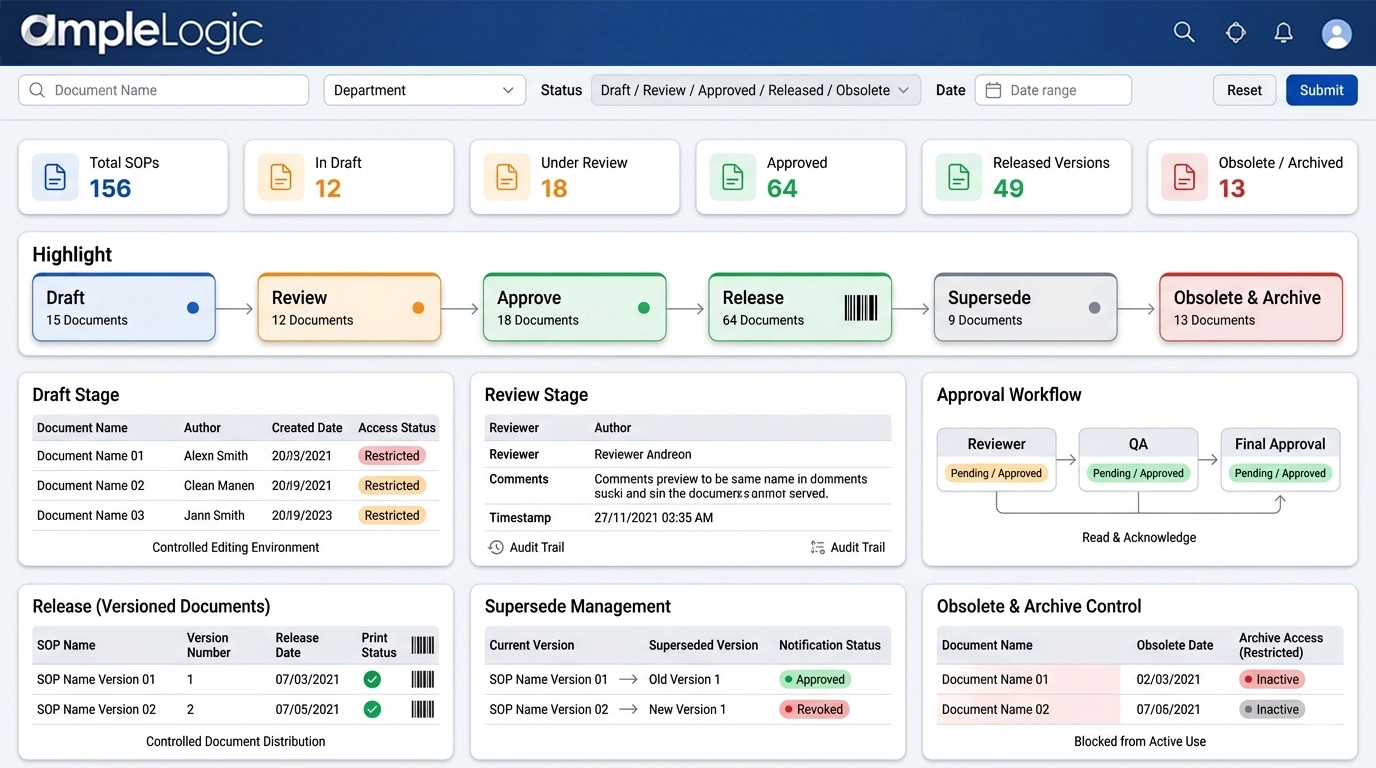
Draft
Controlled document editing with clear draft status and restricted access.
Review
Parallel review by cross-functional teams with complete audit trail of reviewer comments with datetime stamps.
Approve
Formal approval workflow with enforced read and acknowledge step.
Release (Versioned)
Version numbering, print management and barcode assignment for prints.
Supersede
Automated superseding of older versions and notifications to users.
Obsolete and Archive
Explicit revocation of obsolete documents, controlled archive access, and blocked use in active processes.
Draft
Controlled document editing with clear draft status and restricted access.
Review
Parallel review by cross-functional teams with complete audit trail of reviewer comments with datetime stamps.
Approve
Formal approval workflow with enforced read and acknowledge step.
Release (Versioned)
Version numbering, print management and barcode assignment for prints.
Supersede
Automated superseding of older versions and notifications to users.
Obsolete and Archive
Explicit revocation of obsolete documents, controlled archive access, and blocked use in active processes.
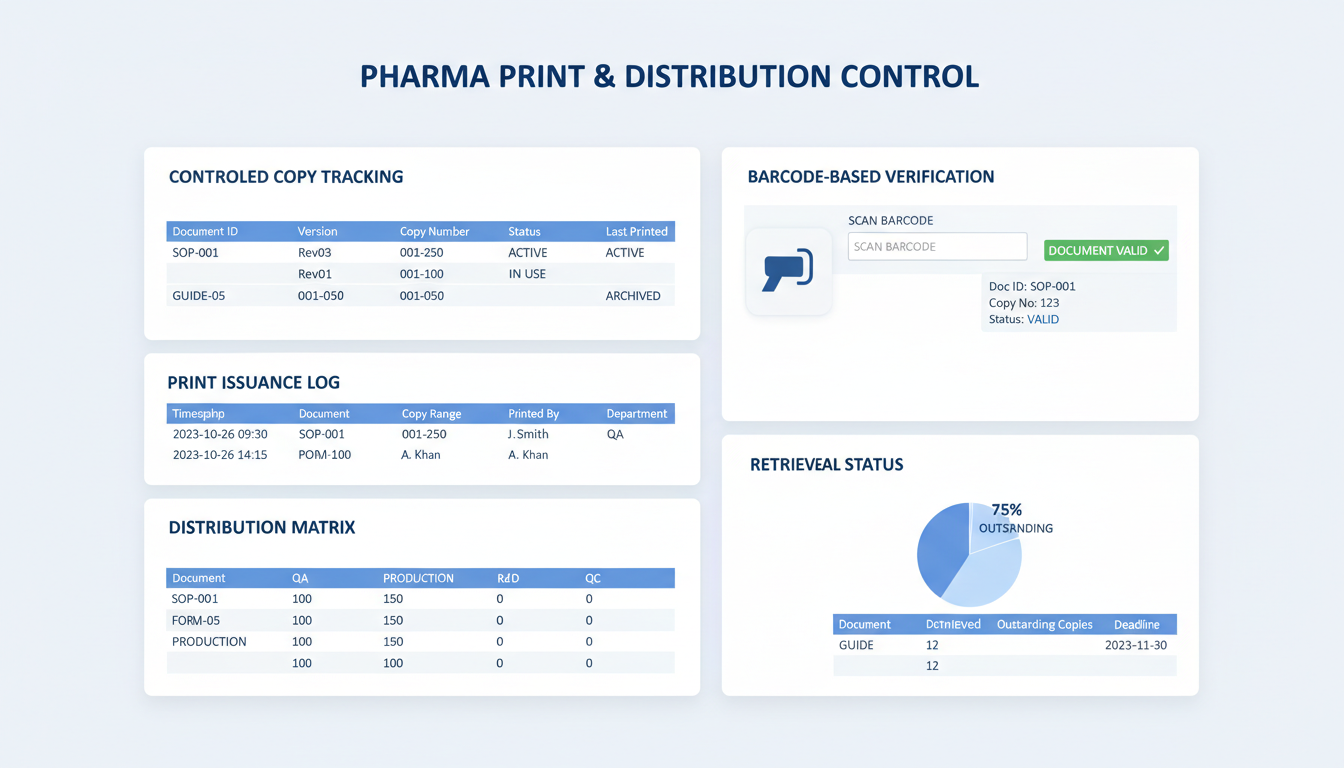
Print Issuance, Controlled Copy Distribution, and Retrieval Tracking
Ensure only current, authorized documents reach production — including uncontrolled, training and draft copies
Approve
Finalize document with version and approval metadata via document approval workflow software.
Lock Version
Freeze approved version; prevent edits and mark superseded states using digital document control system safeguards.
Print Issuance
Grant print rights by role/site, generate watermarks like controlled copy, uncontrolled copy, training copy and draft copy. Enable controlled copies print and retrieval across sites.
Distribution
Publish to controlled site and role-based lists with barcode labels and acknowledgement on handover.
Reconciliation and Audit Trail
Track printing details and reconcile issued paper copies with audits for full pharmaceutical document control system traceability.
Print Control and Verification
Closing the gap between digital systems and physical floor copies through barcode-based tracking
Dynamic Barcode Generation
Automatic watermarking and unique barcode generation for every printed physical copy.
Point-of-Use Verification
Real-time barcode scanning on the production floor to verify document approval status and version.
Comprehensive Print Audit
Full tracking of distributed physical copies including per-user print logs and location data.
Intelligent OCR and AI Search for Your GxP Compliance Document Management System
Make regulated documents searchable, reviewable, and accessible across your digital document control system
AI-Powered OCR
Converts scans and images into searchable, editable text.
Reduce manual transcription and enable faster discovery.
Intelligent Search
Surfaces regulated content with contextual relevance.
Improve retrieval accuracy across document stores.
Voice-Activated Search
Accelerates hands-free retrieval and review.
Faster access during audits and team workflows.
E-Discovery and Audit Support
Enables faster document review, evidence retrieval, and audit responses.
Aid compliance, traceability, and regulated audits.
Team-Wide Accessibility
Provides consistent access to controlled documents across functions.
Enable traceability and collaboration in regulated settings.
GMP-Compliant DMS Integrations with eQMS, LMS, LIMS, and MES
Seamlessly connected to your enterprise GxP document management system ecosystem for end-to-end document traceability
LMS
Training needs from new SOP introduction, version changes. Training completion check.
eQMS
Version change from Change Control, Audit Findings, CAPAs.
RIMS
Version change from regulatory findings.
LIMS
Test methods and STPs with laboratory results and records.
eLog Book
Log book template reference.
Cleaning Validation
Access to SOPs for cleaning procedures.
eBMR/MES
BMR/BPR document management and access.
CAPS
Equipment Manuals/SOPs for calibration procedures.
SAP
Enterprise resource planning integration.
UMS
User management system integration.
AmpleLogic Low-Code DMS Platform Strengths and AI-Enabled Value
Low-code flexibility, compliance readiness, and controlled automation for life sciences
LCNC Drag-and-Drop Designer
Visual builder reduces custom code and speeds delivery for fast app composition.
Cloud Document Management for Pharma
Cloud document management software for pharma with accelerated timelines and quick business impact — ROI in as little as 3 months.
Regulated Industry Document Management
Regulated industry document management software designed for documentation and audits. USFDA, MHRA, ISO ready.
18+ COTS Applications
Prebuilt apps for common life sciences workflows — ready to deploy immediately.
Over 100 Life Sciences Customers
Customer base demonstrates strong industry traction and validated platform adoption.
AI-Enabled Automation
AI assists automation, extraction, and app assembly to accelerate composition and processing.
DMS Software ROI: Business Benefits of Digital Pharmaceutical Document Management
Compliance, speed, accuracy and scalable digital quality
25%
Audit Findings Reduction
Target percent reduction in findings after deployment.
30%
Time to Release Improvement
Projected reduction in time to release documents.
50%
Manual Effort Saved
Estimated reduction in manual processing hours.
"Easy to access and handle. Paperless work and can keep records of change control taken and ease to access from every location. Less time consumable."
Shweta Ramdas R.
Document Control Specialist
"The software is easy to use and implement at operator level. Online activity with audit trails makes compliance seamless."
Silpa K.
Quality Assurance
"Configurable workflows, compliance benefits and reduction of paper usage across the organization. The customer support is prompt and implementation was straightforward."
Ramesh T.
Pharma IT Manager
Schedule a Live DMS Demo with Sample SOPs and Batch Records
Experience controlled printing, print issuance, and end-to-end traceability with AmpleLogic's AI-powered electronic document management system.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.