Go Paperless — Real-Time Batch Manufacturing With eBMR eBMR
AmpleLogic eBMR/MES digitizes batch manufacturing with real-time shop floor execution, equipment integration, and review by exception -- cutting batch release time by 50% while maintaining full 21 CFR Part 11 compliance.
50%
Faster Batch Release
Zero
Paper on Shop Floor
99%
Right-First-Time Rate
10x
Faster Deployment
Why Paper Batch Records Are Holding Back Pharma Manufacturing Compliance
Manual Batch Records and Legacy MES Systems Create Systemic GMP Risks Across Manufacturing Operations -- From Compliance Exposure to Production Delays.
Human Errors & Manipulation Risk
Manual paper records are prone to transcription mistakes, missed entries, and data manipulation — pharma MES software and electronic batch manufacturing eliminate these risks through batch manufacturing automation.
Error-Prone Batch Data
Product and batch data recorded on paper lacks real-time production monitoring, leading to specification failures and rework that pharmaceutical manufacturing software prevents.
Lack of Real-Time Visibility
Paper-based systems provide no live production status — a digital manufacturing platform with real-time production monitoring and EE monitoring replaces delayed reports and walk-throughs.
Challenging Audits & Inspections
Retrieving, compiling, and verifying paper records during FDA/MHRA inspections is time-consuming — GxP compliant MES and GMP manufacturing software ensure instant audit readiness.
Compliance & Security Gaps
Paper lacks robust access controls, tamper-proof audit trails, and electronic signatures required by 21 CFR Part 11 — this life sciences MES platform closes all compliance gaps.
Limited Collaboration & Delays
Sequential paper hand-offs between departments slow batch execution — automated pharma regulatory workflows and shopfloor execution system capabilities eliminate bottlenecks.
Unreliable Material Tracking
Manual material logs lead to discrepancies — weighing scale integrations with barcode verification ensure accurate dispensing records and yield reconciliation.
Difficult Standards Maintenance
Keeping paper SOPs and master records current is operationally costly — recipe management with low-code manufacturing applications simplifies updates across products and sites.
Complete Electronic Batch Manufacturing Record & MES -- One GMP-Compliant Platform
From Master Batch Recipe Creation Through Batch Execution, Quality Checks, and Release -- Every eBMR Module Works Together on a Single GMP Data Layer.
Execution & Real-Time Production Monitoring
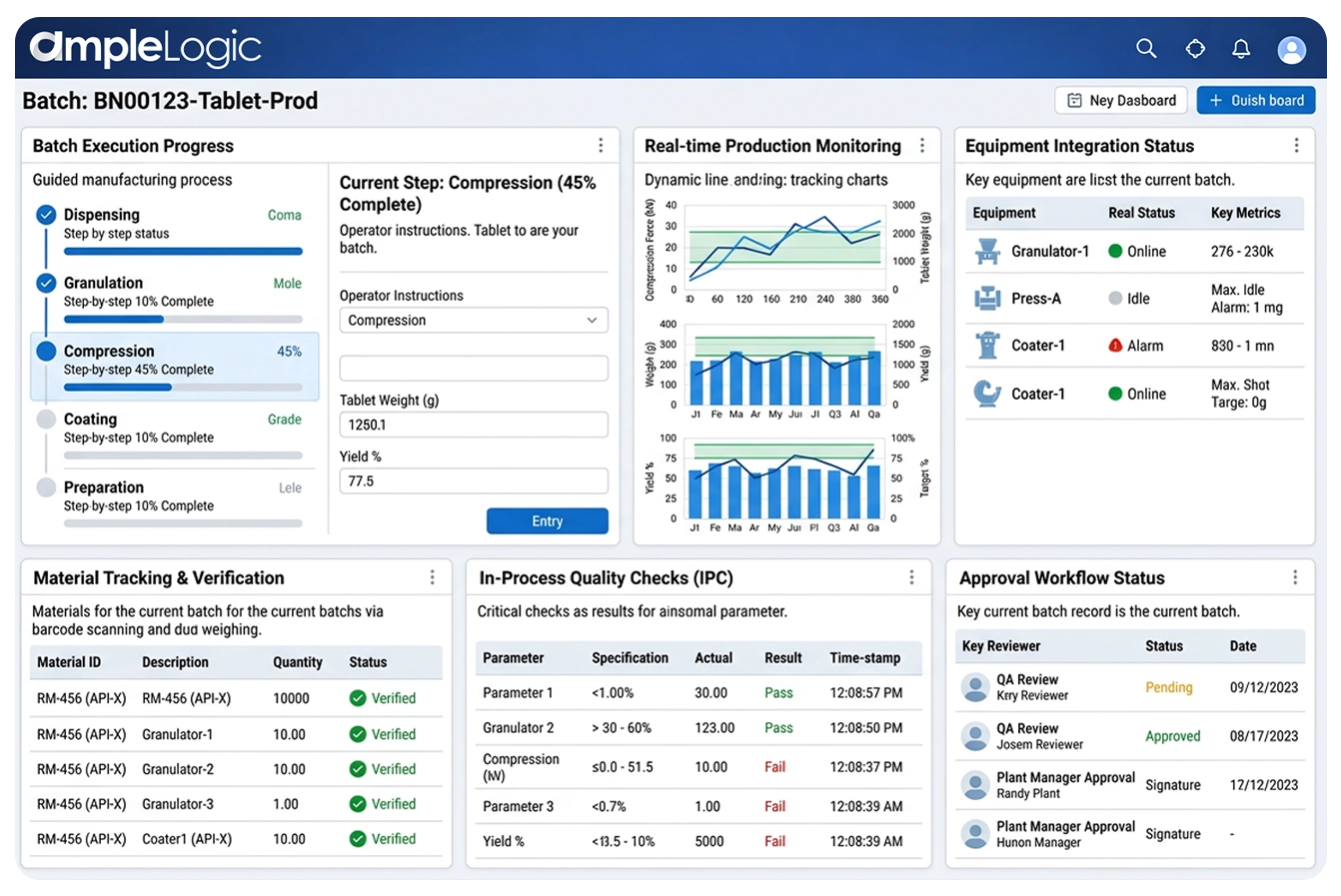
Shopfloor execution system with real-time production monitoring, step-by-step guided processes, live production dashboards, EE monitoring, and automated data capture from equipment on this pharma MES software.
Enterprise-Grade Pharmaceutical Manufacturing Execution Capabilities
Production Monitoring, Equipment Integration, and GMP-Compliant Workflows — Every Capability for a Paperless Pharma Manufacturing Floor.
Real-Time Data Capture & Production Monitoring
Pharma MES software with automated data capture from equipment, PLCs, and instruments — real-time production monitoring and EE monitoring eliminates manual transcription on this digital manufacturing platform.
Equipment Integration (PLC/HMI/SCADA)
Shopfloor execution system with direct integration via OPC, Historian databases, and standard PLC protocols for real-time process data on this smart factory platform.
Electronic Signatures & Audit Trails
GxP compliant MES with 21 CFR Part 11 compliant e-signatures, immutable time-stamped audit trails for every action, change, and approval in this GMP manufacturing software.
Automated Workflow Management
Batch manufacturing automation with configurable multi-level approval workflows, automatic routing, escalation, and parallel/sequential processing on this pharmaceutical manufacturing software.
Review by Exception
AI-driven manufacturing insights highlighting only exceptions and deviations for QA review, dramatically accelerating batch release on this life sciences MES platform.
Reminders & Escalation Alerts
Cloud MES platform with automated email alerts for pending tasks, batch approval stages, overdue activities, and escalation to management.
Product Label Management
Digital manufacturing platform with centralized label management ensuring consistency, version control, and regulatory compliance of all product labels.
Packaging Components
Pharmaceutical manufacturing software handling diverse packing requirements per product with configurable bill-of-materials, component tracking, and reconciliation.
Low-Code Manufacturing Applications
Low-code manufacturing applications with intuitive drag-and-drop interface for tailoring processes — enabling pharma manufacturing digitalization without custom coding.
Advanced Production Scheduling
Smart factory platform with Industry 4.0 manufacturing scheduling algorithms optimizing resource allocation, minimizing changeover time, and maximizing throughput.
Scalable Cloud MES Platform
Cloud MES platform that is 100% customizable — scale from a single line to enterprise-wide multi-site deployment for biopharma manufacturing platform needs.
Complete Production Lifecycle Visibility
Digital manufacturing transformation with end-to-end visibility and control across the entire production lifecycle from planning through batch release and archival.
Drug Product Batch Release Simplified
A Structured End-to-End eBMR Flow That Guides Your Pharmaceutical Manufacturing Process from Recipe Creation to Final Batch Approval.
Master Setup
- Creation of masters (products, materials, equipment, rooms)
- Product & stage mapping
- Mapping of raw materials to products
- Room & equipment mapping
- Sequential & parallel process definition
Recipe & Scheduling
- Master recipe creation (drag-and-drop)
- Production scheduling & resource allocation
- Batch planning and issuance
Batch Execution
- Dispensing of raw materials with barcode verification
- Step-by-step guided batch execution
- Equipment integration & auto data capture
- In-process quality checks & test results
- Room & equipment log entries
Review & Release
- Exception handling & deviation management
- Electronic signatures & multi-level approvals
- Review by exception for faster batch release
- Complete audit trail & batch genealogy
Stepwise Migration: Paper to Full eBMR
BIMS (Batch Record Issuance Management)
- Master copy version management and document issuance
- Auto printing to eliminate manual mistakes
- Mother batch, co-pack, and bilayer logic
- Quantity management (debit & credit)
- Control on manual recording mistakes, additional page requests, ratifications
eBatch Recording
- Issuance process converted to online workflow
- Record batch information online (move from hardcopy)
- Measures to confirm recording is done at equipment only
- Integration with eQMS systems
Full eBMR
- Move from manual recording to auto-populate data from equipment
- Real-time data integration with PLCs, HMI, and SCADA
- Complete paperless manufacturing floor
- Full 21 CFR Part 11 / EU Annex 11 compliance
Simplifying Pharma Shop Floor Execution with Electronic Batch Records
Purpose-Built for Pharma Operators on the GMP Manufacturing Floor -- 100% Compliance, Automated Validation.
Guided Processes
Shopfloor execution system with user-friendly step-by-step instructions for executing manufacturing steps — operators follow intuitive prompts ensuring consistency on this pharma MES software.
Real-Time Production Monitoring
Real-time production monitoring with animated visual cues and status icons helping operators understand current activity status at a glance with EE monitoring on this digital manufacturing platform.
Auto Validations
Batch manufacturing automation with system warnings for incorrect entries, out-of-spec values, and missed steps preventing errors before they reach the electronic batch manufacturing record.
On-Screen SOPs
GMP manufacturing software displaying Standard Operating Procedures directly on-screen at the point of use for immediate reference during execution.
Activity Guidance & Next Steps
Smart factory platform with clear instructions for the next steps in each task, ensuring operators always know what comes next in the process on this life sciences MES platform.
Barcode & QR Scanning
Weighing scale integrations and integrated barcode/QR scanning for material verification, equipment identification, and error-proof data entry on the Industry 4.0 manufacturing shop floor.
Seamlessly Integrated eBMR/MES Across Your Entire Pharma Operation
Native eBMR/MES Integration Across Quality, Lab, Training, Equipment, and Enterprise Systems -- All on One 21 CFR Part 11 Compliant Platform.
Quality Systems
- eQMS (CAPA, Deviations, Change Control)
- OOS/OOT Management
- Audit Management
- Complaint Handling
Laboratory
- LIMS (In-Process Testing, CoA)
- Stability Studies
- Environmental Monitoring (EMS)
Document & Training
- DMS (SOPs, Master Records)
- LMS (Operator Training, Competency)
- Electronic Logbooks (eLogs)
Equipment & Automation
- PLC / HMI / SCADA Integration
- OPC Server Connectivity
- Historian Database
- Weighing & Dispensing Systems
Enterprise Systems
- SAP / Oracle ERP
- Warehouse Management (WMS)
- Supply Chain Management
- Financial Systems
Calibration & Maintenance
- CAPS (Calibration Scheduling)
- Preventive Maintenance
- Equipment Qualification
- Asset Lifecycle
How AmpleLogic eBMR/MES Compares to Traditional Pharma MES Solutions
See How AmpleLogic eBMR Stacks Up Against Traditional, Enterprise, and Automation-Centric Pharmaceutical MES Solutions.
| Feature | AmpleLogic | Cloud MES | Enterprise MES | Automation MES |
|---|---|---|---|---|
| Deployment Speed | Weeks (low-code aPaaS) | Months (cloud SaaS) | 6-18 months (on-prem/cloud) | 6-12 months (on-prem/cloud) |
| Customization | 100% drag-and-drop, no coding | Template-based configuration | Professional services required | Requires system integrators |
| Recipe Management | Visual drag-and-drop builder | Template-driven EBR | Recipe-driven ISA-88 engine | Recipe management module |
| Equipment Integration | PLC/HMI/SCADA via OPC | Limited (QMS-focused) | Deep ISA-95 / OPC-UA | Vendor-locked PLC ecosystem |
| Quality System Integration | Native (same platform) | Native QMS + EBR | Third-party integration | Third-party integration |
| Unified Platform | 14+ apps on single aPaaS | QMS + Manufacturing only | MES-only (closed ecosystem) | MES-only (vendor ecosystem) |
| Infrastructure Cost | Up to 70% lower (open-source) | Cloud subscription model | High (enterprise licensing) | High (platform + integrators) |
| Continuous Product Upgrades | Included, regular releases | Cloud auto-updates | Version upgrades (project-based) | Major version releases |
100% Customizable via Low-Code
Low-code manufacturing applications — every workflow, form, and process fully configurable to match your exact manufacturing requirements without custom coding on this pharma MES software.
Drag-and-Drop Recipe Builder
Recipe management with visual recipe creation tool letting process engineers build and modify master recipes without IT involvement on this digital manufacturing platform.
Cloud MES Platform with Continuous Upgrades
Cloud MES platform with regular product releases, new features, regulatory updates, and performance improvements — enabling digital manufacturing transformation included in your subscription.
Faster Deployment for Industry 4.0
Smart factory platform and life sciences MES platform enabling deployment in weeks — not the 6-18 months typical of traditional MES implementations — accelerating pharma manufacturing digitalization.
The Business Case for Paperless Pharmaceutical Manufacturing
Quantified Results — Accelerate Batch Release, Reduce Paper Costs, and Improve Manufacturing Accuracy After Deploying AmpleLogic Electronic Batch Manufacturing Record Software.
Faster Batch Release
Review by exception and automated workflows cut batch release time in half compared to paper-based processes.
Reduction in Paper Costs
Eliminate paper batch records, printing, storage, and retrieval costs across your manufacturing operations.
Fewer Data Entry Errors
Auto-validation, equipment integration, and guided processes virtually eliminate manual transcription errors.
Lower Infrastructure Costs
Open-source infrastructure stack (Linux, PostgreSQL) dramatically reduces IT infrastructure spend.
Return on Investment
Customers typically achieve 3-5x ROI within the first 18 months of deployment through operational efficiencies.
Faster Audit Preparation
Instant access to immutable electronic records with complete audit trails accelerates inspection readiness.
eBMR/MES Compliant with 16+ Global Pharmaceutical Regulatory Bodies
Built for Pharmaceutical Regulatory Compliance from Day One -- FDA 21 CFR Part 11, EU Annex 11, GAMP 5, and More.
Compliance Standards
Regulatory Bodies
USFDA
USA
MHRA
UK
TGA
Australia
CDSCO
India
Health Canada
Canada
MCC
South Africa
ANVISA
Brazil
EMEA
European Union
SFDA
China
MPA
Sweden
Swissmedic
Switzerland
MCAZ
Zimbabwe
MEDSAFE
New Zealand
MHLW
Japan
NAFDAC
Nigeria
KFDA
Korea
Purpose-Built for Regulated Manufacturing
Pharmaceutical Manufacturing
- Oral Solid Dosage
- Liquid & Semi-Solid
- Sterile Injectables
- API Manufacturing
- Pharma Manufacturing Digitalization
Biopharma Manufacturing Platform
- Biologics Production
- Cell Culture
- Purification
- Fill & Finish
- Biotech Manufacturing Execution
Medical Devices
- Device Assembly
- Packaging & Labeling
- Sterilization Records
- DHR Management
CDMO / CMO
- Multi-Client Batch Records
- Campaign Manufacturing
- Technology Transfer
- Rapid Site Onboarding
- Digital Manufacturing Transformation
"Production activities are now very easy and stable. The transition from paper BMRs to electronic has saved us significant time and reduced errors."
Production Manager
Mid-Market Pharmaceutical
"Makes issuance and tracking of BMRs much easier. The paperless workflow streamlined our entire batch documentation process."
QA Specialist
Enterprise Manufacturing
"Easy to implement and the support team guided us through the entire process. Integration with our document management system works seamlessly."
IT Director
Pharmaceutical Company
Ready to Eliminate Paper from Your Shop Floor?
See how AmpleLogic eBMR/MES can digitize your batch manufacturing and cut release time by 50%. Schedule a personalized demo today.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.