Replace Paper Logbooks With ALCOA+ Compliant Digital Logs
AmpleLogic is a GMP compliant electronic logbook system purpose-built for pharma production logbook software needs. Close the operational context gap between legacy equipment and modern MES through AI-powered paperless logbooks in pharmaceutical manufacturing — digitize observations without impacting validated system logic.
100%
Audit Trail Depth
#1
Regulatory Alignment
100%
E-Signature Adoption
10x
Faster Deployment with AI
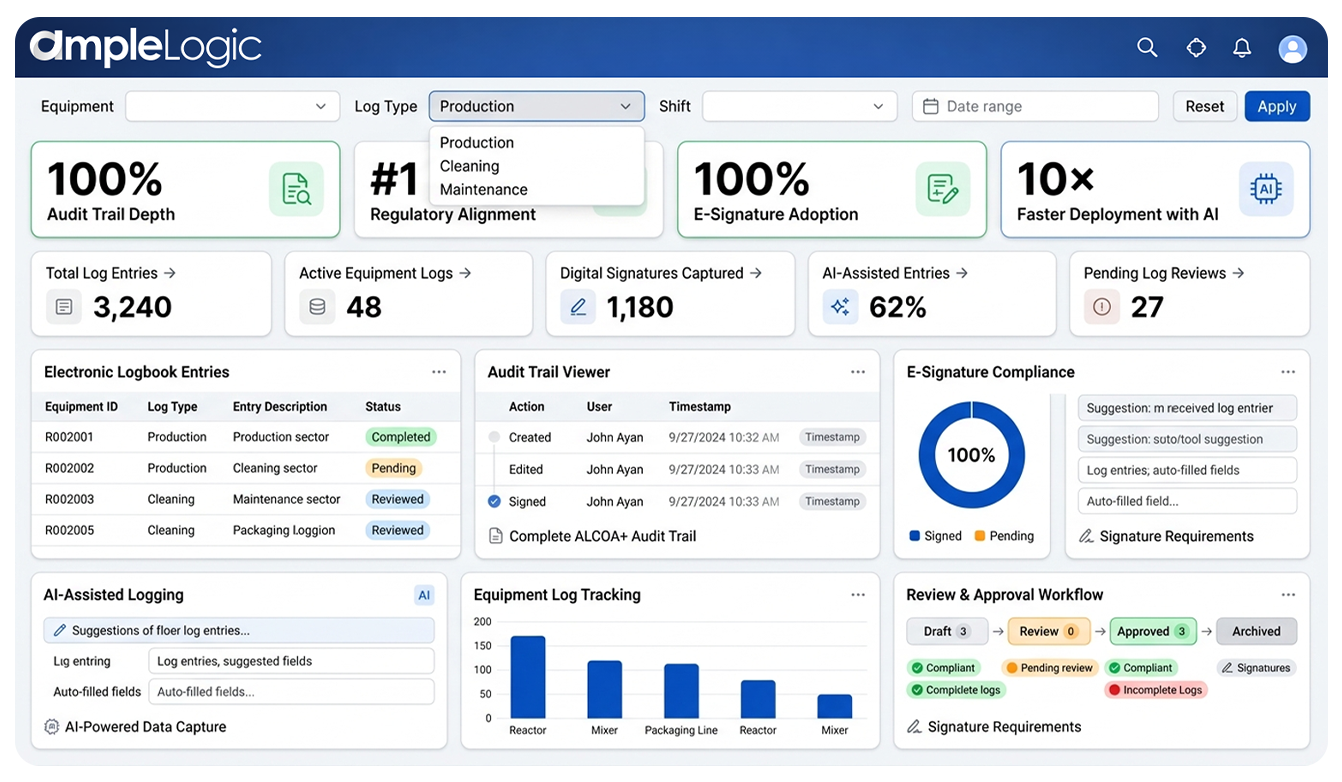
21 CFR Part 11 & EU Annex 11 Compliant Electronic Logbook for Pharma
eLogbook architecture is engineered to satisfy the most rigorous regulatory expectations for electronic records and data integrity
Audit Trail Depth
Full capture of every entry, modification, and deletion with timestamps. ALCOA+ attributable and contemporaneous records from day one.
Regulatory Alignment
Fully compliant with FDA 21 CFR Part 11 and EU Annex 11 frameworks. Built to satisfy the most rigorous GxP expectations globally.
E-Signature Adoption
Enforced biometric or MFA authentication for all critical GxP approvals. Non-repudiation and signature linking per Part 11 requirements.
Data Integrity by Design
Every record is attributable, legible, contemporaneous, original, and accurate. Completeness, consistency, endurance, and availability are enforced automatically.
eLogbook: The Flexible Equipment Operations and Offline Recording for Life Sciences Operations
eLogbook serves as the contextual bridge above both automated MES execution and raw instrumentation data
MES Execution Layer
Structured transactional data and batch record enforcement for high-speed automated lines. Handles binary pass/fail transactions and structured manufacturing workflows.
eLogbook Context Layer
eLogbookCaptures observational data, situational judgments, and exception-based operational context. The flexible documentation layer above both automated and manual shop floor activities.
Instrumentation Layer
Raw data from PLCs, SCADA, and standalone legacy equipment across the facility. Real-time equipment readings, sensor data, and automated alarms feed into the eLogbook.
Why MES Alone Is Not Sufficient for Pharma GMP Documentation Compliance
Transactional systems prioritize execution speed, often losing the vital human context of operations
Indirect Equipment Blind Spots
MES lacks the flexibility to document indirect equipment like HVAC or Purified Water systems without heavy customization. eLogbooks capture these critical observations natively.
Missing Human Context
ELBs capture operator assessments and situational judgments that are not binary pass/fail transactions. The human context of operations is preserved for audits and investigations.
Unstructured Activity Gaps
Documentation of unstructured activities like shift handovers and exception narratives is natively supported in eLogbooks versus structured MES logic.

The eLogbook Advantage
MES vs. Electronic Logbook: GMP Documentation Scope Comparison
Comparing the scope of documentation across Manufacturing Execution and Logbook layers
| Activity Category | MES Focus | eLogbook Focus |
|---|---|---|
| Cleaning & Clearance | Pass/Fail Timestamp | Visual observations & specific methods used |
| Utility Monitoring | Automated Alarms | Situational narrative of deviations (e.g. HVAC) |
| Shift Transitions | N/A (Process specific) | Continuity narrative and institutional knowledge |
| Calibration | Reference date | Verification logs and manual instrument checks |
| Environmental Monitoring | Automated sensor logs | Operator observations, deviation narratives, corrective notes |
| Material Handling | Barcode scan events | Receipt conditions, visual inspections, storage exceptions |
GMP Equipment Logbook Software for Pharma, Biotech & Regulated Manufacturing
From drag-and-drop logbook builder to cleanroom logbook software and warehouse logbook software — every capability designed for GxP-regulated environments
Real-Time Data Integration
Live feeds from PLCs, SCADA, and standalone instruments stream directly into the electronic logbook for pharmaceutical manufacturing. Eliminate manual transcription errors and ensure contemporaneous data capture across the facility.
Drag-and-Drop Logbook Builder
Equipment Logbook Software for Pharma
Cleanroom Logbook Software
Calibration Logbook Software
QC Lab Logbook Software
Environmental Monitoring Logbook
Warehouse Logbook Software
Role-Based Logbooks
Checklists & Interlocking
Smart Notifications & Alerts
Analytics & Dashboards
QR / Barcode Scanning
Offline Access (Native App)
Voice-Enabled Logging
Immutable Audit Trail
Smart Report Generation
E-Signature Workflows
Electronic Logbook Across 4 Pharmaceutical Operational Pillars
Consolidated automated logging domains within the AmpleLogic eLogbook ecosystem
Area & Equipment Logs
- Monitor AHU, Chiller, and Boiler operations with equipment logbook software for pharma
- Automate calibration logbook software and Preventive Maintenance records
- Track equipment maintenance logbook lifecycle, status changes, and utilization
- Record environmental conditions using environmental monitoring logbook for classified areas
QC Laboratory Operations
- Bridge the LIMS gap with QC lab logbook software for physical inspections and sanitization
- Track column lifecycle and instrument usage logs
- Record reagent preparation, standardization, and expiry
- Document media preparation and growth promotion testing
Production & Warehouse
- Digitize Dispensing, Packing, and Coating logs with pharma production logbook software
- Warehouse logbook software for material handling and environmental records
- Track in-process controls and line clearance activities
- Document batch start/stop, yield reconciliation, and exceptions
Quality & Compliance
- Automate OOC, Extension Requests, and Deviation logs
- Record training attendance and material issuance logs
- Manage change control narratives and impact assessments
- Track CAPA actions linked to logbook observations

Implementation Roadmap
Legacy Assessment
Identify high-risk paper logs and aging equipment lacking digital output. Map critical documentation gaps and prioritize areas for maximum compliance impact.
Non-Intrusive Overlay
Deploy eLogbook templates without modifying underlying control logic or validated PLC/SCADA code. Avoid costly re-validation cycles with read-only integration.
Validation & Training
Execute 21 CFR Part 11 validation protocols and train operators on digital observation capture. Comprehensive IQ/OQ/PQ documentation included.
Enterprise Synergy
Integrate eLogbook data with MES, ERP, eQMS, and LIMS for unified operational reporting. Achieve a single source of truth across the manufacturing enterprise.
AI-Driven Auto Generation of GxP Logs from Word Templates to Reduce Documentation Time
AmpleLogic's low-code engine converts standard Word templates into fully compliant, version-tracked electronic records instantly — AI driven auto generation of logs from word templates eliminates manual configuration entirely
AI-Driven Auto Generation of Logs from Word Templates
Upload any existing Word template; the AI automatically identifies data structures and maps compliance fields without manual effort. AI driven auto generation of logs from word templates converts paper forms to digital eLogbooks instantly — eliminating weeks of manual configuration.
Automated Compliance
Generated eLogbooks inherently meet FDA 21 CFR Part 11 and EU Annex 11 standards. This GMP compliant electronic logbook system delivers global audit readiness from day one with zero manual compliance configuration.
Seamless Versioning
Built-in tracking captures every change across the document lifecycle. Provides a transparent and immutable audit trail with full traceability of who changed what and when.
Rapid Deployment (10x Faster)
Achieve go-live status 10x faster than traditional methods. AI dramatically reduces the time business teams spend on configuration, validation, and template creation.
Anomaly Detection
AI spots data outliers to identify errors, compliance issues, or out-of-specification conditions in real time. Proactive alerts prevent deviations before they escalate.
Predictive Maintenance
AI predicts equipment failures using historical logbook data and sensor trends. Automated alerts prevent unplanned downtime and ensure compliance with maintenance schedules.
Cost-Effective Electronic Logbook Alternative to Custom Pharma Software Integrations
eLogbooks eliminate the need for tailor-made hardware interfaces for every piece of shop floor equipment
Custom Hardware Interfaces for Legacy Equipment
Custom hardware & point integrations
No custom hardware; non-intrusive ELB
Deployment Timelines
Long MES programs (years)
Rapid ELB rollout (months)
Lifecycle Maintenance & Upgrades
Tightly coupled systems
Decoupled ELB architecture
A Pathway to Digital Maturity
Pragmatic Digitization
Replace paper logs with eLogbooks for immediate compliance wins and data integrity. Achieve audit readiness without disrupting validated systems.
Template Standardization
Unify documentation across shifts, sites, and departments to ensure institutional knowledge retention and consistent GxP practices globally.
Integrated Context
Synergize eLogbook data with MES metrics for a holistic 360-degree view of the batch. Enable predictive analytics and continuous improvement.
Digitizing Legacy GxP Environments with Regulatory Compliant Electronic Logbook Software
Non-intrusive integration with standalone instruments, PLCs, SCADA systems, and enterprise applications
eQMS
Automated deviation, CAPA, and change control workflows linked to logbook observations
LIMS
Synchronize lab results, instrument logs, and QC sample tracking in real time
ERP & Maintenance
Integrate equipment status, spare parts, and preventive maintenance schedules
eBMR / MES
Bidirectional batch context sharing between execution records and observational logs
PLC / SCADA / Data Logger
Non-intrusive read-only connections via RS 232/485, Ethernet, API, or OPC protocols
Third-Party Applications
Open API and CAMS/OPC/Link Server connectivity for any external system integration

Equipment Logic Integrity
Achieve digitization without modifying validated PLC or SCADA code. Read-only, non-intrusive connections avoid costly re-validation cycles while capturing complete operational context.
Industries Using AmpleLogic Electronic Logbook for GMP Pharmaceutical Manufacturing
GxP-compliant electronic logbook solutions — from cleanroom logbook software and QC lab logbook software to warehouse logbook software — tailored for regulated manufacturing environments
Pharmaceuticals
End-to-end electronic logbook for pharmaceutical manufacturing — digitize R&D, API, formulation, and finished dose production with GMP compliant electronic logbook system capabilities.
Biotechnology
Specialized paperless logbooks in pharmaceutical manufacturing for bioprocessing, fermentation, cell culture, and biologics production environments.
Medical Devices
Compliant cleanroom logbook software for medical device manufacturing, sterilization, assembly, and packaging operations.
Food & Beverages
HACCP-aligned digital logbooks for production, sanitation, CIP/COP, and environmental monitoring logbook workflows in food manufacturing.
Gene Therapy
Precision logging for viral vector production, cell therapy manufacturing, and cold chain management with electronic equipment logbook pharma compliance.
Cosmetics & Beauty
GMP-compliant electronic logbooks for batch manufacturing, quality control, and hygiene monitoring powered by pharma production logbook software.
"The compliance and audit trail features are excellent. It supports FDA 21 CFR Part 11 and EU Annex 11, giving us full confidence in our electronic records."
Verified User
Quality Assurance, Pharma
"Harmonization of logs across departments is seamless. Equipment usage tracking and integration with our eDMS and LMS made adoption very smooth."
IT Manager
Mid-Market Pharmaceutical
"Implementation was fast and the support team is very responsive. The web-based UI makes daily logbook tasks simple for our operators."
QC Supervisor
Enterprise Pharmaceutical
Achieve 100% Audit Readiness with Paperless Logbooks
Join the leading life sciences companies using AmpleLogic electronic logbook for pharmaceutical manufacturing to eliminate manual compliance risks and accelerate digital transformation with AI-powered paperless logbooks in pharmaceutical manufacturing.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.