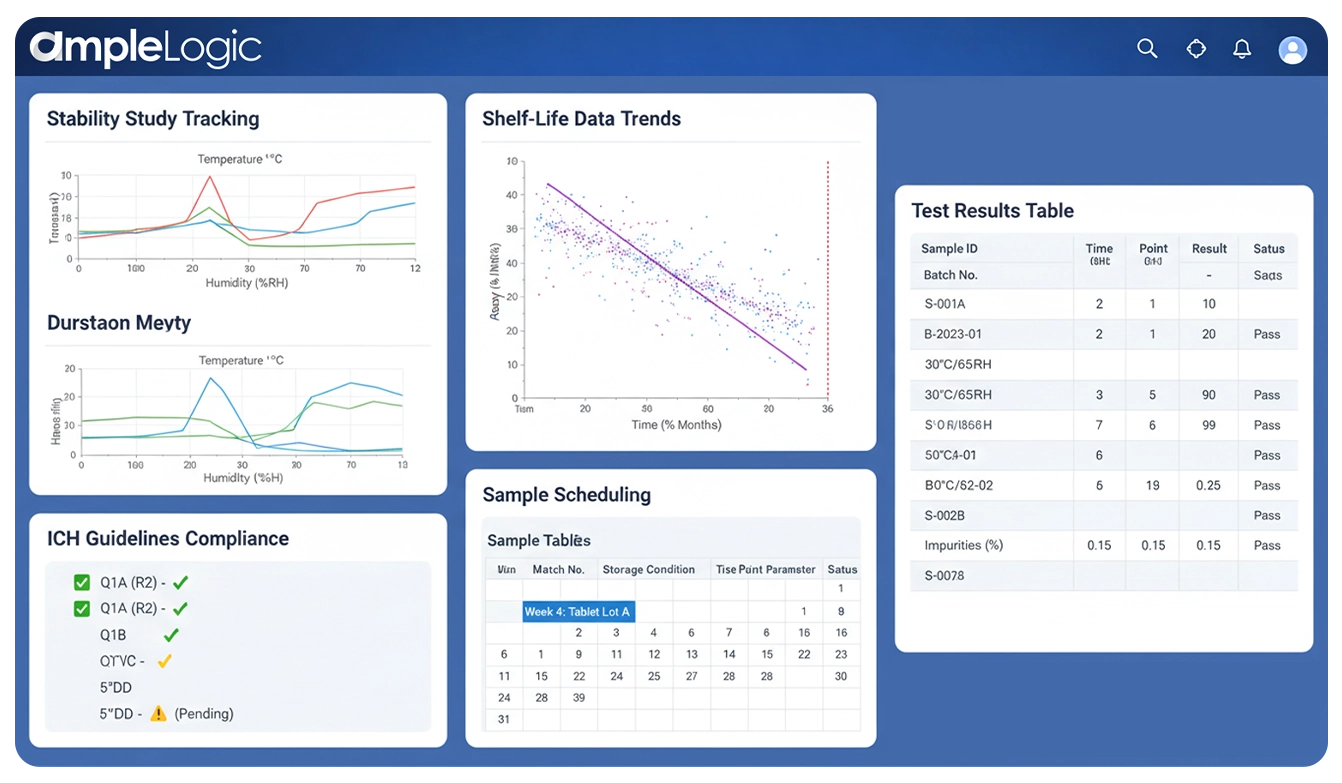
Manage Stability Studies End to End Inside Your LIMS
AmpleLogic delivers end-to-end stability schedule management — from stability test protocol creation and stability schedule calendar planning through chamber monitoring, test execution, trend analysis, shelf-life determination, and regulatory-ready reporting aligned to ICH Q1A, FDA, and GMP requirements.
40%
Faster Study Setup
100%
ICH Compliance
30%
Reduced Missed Pulls
60%
Less Manual Tracking
50%
Cost Savings
3–6
Month Deployment
What Is Stability Schedule Management?
Stability schedule management is the systematic planning, execution, and tracking of pharmaceutical stability studies. It ensures every product batch undergoes the correct storage conditions, sampling time points, and analytical testing as defined by the stability test protocol — enabling accurate shelf-life determination and regulatory compliance with ICH Q1A, FDA 21 CFR Part 211.166, and GMP requirements.
Stability Test Protocol
A stability test protocol defines the study design — storage conditions, time points, test parameters, acceptance criteria, and packaging configurations. AmpleLogic automates protocol creation with ICH Q1A-compliant templates.
Stability Schedule Calendar
The stability schedule calendar is a visual timeline of all pull dates, chamber allocations, and analyst assignments across every active study. AmpleLogic auto-generates and maintains this calendar in real time.
Stability Schedule Management
Stability schedule management coordinates the entire lifecycle — from protocol approval through sample pulls, test execution, trend analysis, and shelf-life determination — in a single validated platform.
Challenges in Stability Schedule Management Without Automation
Organizations relying on manual processes for stability test protocol management and stability schedule calendar tracking face critical risks.
Manual Stability Schedule Management
Teams rely on spreadsheets and paper logs to plan stability schedules, leading to missed pull points, scheduling conflicts, and lack of real-time visibility into study timelines.
Inconsistent Stability Test Protocols
Without a standardized system, stability test protocol creation varies across teams and sites, causing regulatory gaps, audit findings, and inconsistent study designs.
No Centralized Stability Schedule Calendar
Absence of a unified stability schedule calendar means analysts cannot see upcoming pulls, chamber allocations, or overlapping studies in one place.
Disconnected Environmental Data
Chamber temperature and humidity data is tracked separately from stability studies, creating traceability gaps and delayed excursion response.
Delayed Trend Analysis & Reporting
Manual data compilation for stability trend analysis delays shelf-life determination, regulatory submissions, and product launch timelines.
Compliance Risk from Fragmented Systems
Using multiple disconnected tools for stability program management increases data integrity risks and makes ICH Q1A and FDA audit readiness difficult.
AmpleLogic Approach to Stability Test Protocol & Schedule Automation
Six integrated steps that transform manual stability schedule management into a fully automated, ICH-compliant workflow.
Define Stability Test Protocol
Create ICH Q1A-compliant stability test protocols with configurable study designs, storage conditions, acceptance criteria, and automated sampling schedules — all in a single validated system.
Build Stability Schedule Calendar
Auto-generate a visual stability schedule calendar with time-point pull dates, chamber assignments, analyst allocation, and automated reminders — eliminating manual planning entirely.
Monitor Chamber Conditions
Real-time temperature and humidity monitoring with automated excursion alerts, sensor integration, and environmental data linked directly to stability study records.
Execute & Capture Results
Run analytical tests within the integrated LIMS workflow with direct instrument connectivity, in-line calculations, and full sample-to-result traceability.
Analyze Trends & Determine Shelf-Life
AI-powered stability trend analysis with degradation modeling, statistical shelf-life projection, expiry determination, and confidence interval reporting.
Generate Regulatory Reports
Produce ICH-compliant stability reports, FDA submission packages, and GMP stability program documentation automatically from your stability data.
AmpleLogic Stability Schedule Calendar & Data Hub
A centralized platform that unifies stability test protocol management, stability schedule calendar, trend analytics, and compliance documentation in one place.
Unified Study Dashboard
Single view of all active, completed, and planned stability studies with real-time status, upcoming pull dates, and overdue alerts across every product and condition.
Stability Schedule Management Engine
Centralized stability schedule management with automated pull scheduling, dynamic rescheduling, priority-based queue management, and calendar-based views for every study.
Interactive Trend Analytics
Multi-batch overlay charts, degradation curve fitting, OOT/OOS detection, and predictive shelf-life forecasting — all accessible from a single analytics console.
Complete Audit Trail
Every action — from stability test protocol creation through result entry and report generation — is captured with timestamped, user-identified, ALCOA+ compliant audit records.
Smart Notification Center
Configurable alerts for upcoming pulls, overdue tests, OOT results, chamber excursions, and protocol deviations with multi-level escalation workflows.
Cross-System Integration
Bi-directional integration with LIMS, ERP, QMS, and instrument systems to create a single source of truth for all stability data across your organization.
Manual vs. Automated Stability Schedule Management
See how AmpleLogic transforms every aspect of stability test protocol management and stability schedule calendar operations.
| Area | Before (Manual) | After (AmpleLogic) |
|---|---|---|
| Study Scheduling | Manual spreadsheet tracking, scheduling conflicts | Automated stability schedule calendar with reminders |
| Protocol Creation | Word documents, inconsistent formats across sites | Standardized stability test protocol templates (ICH Q1A) |
| Pull Management | Missed pulls, no real-time visibility | Zero missed pulls with auto-scheduling & alerts |
| Chamber Monitoring | Disconnected sensors, delayed excursion response | Real-time monitoring with instant alerts |
| Trend Analysis | Manual Excel charting, weeks to compile | AI-powered analysis, instant shelf-life projection |
| Reporting | Manual compilation, error-prone | One-click ICH/FDA regulatory-ready reports |
| Audit Readiness | Scramble to gather evidence | Always audit-ready with complete ALCOA+ trail |
Trusted Across Life Sciences
Regulatory Compliance for Stability Test Protocol & Schedule Management
Ready to Automate Your Stability Schedule Management?
See how AmpleLogic transforms stability test protocol creation, stability schedule calendar planning, and shelf-life determination into a single automated, ICH-compliant workflow.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.