Monitor Process Performance in Real Time With CPV
AmpleLogic transforms continued process verification from manual statistical monitoring into an AI-driven manufacturing intelligence platform that predicts deviations, optimizes processes, and ensures consistent product quality.
Real-Time
CPP/CQA Monitoring
75%
Effort Reduction
100%
Audit Trail
6σ
Statistical Analysis
Current State: Manual CPV Challenges in Pharma Process Validation
Operational bottlenecks that delay trend detection, increase rework, and raise audit risk in continued process verification
Manual Data Collection from Multiple Systems
CPP and CQA data scattered across LIMS, MES, ERP, and spreadsheets. Manual aggregation is time-consuming, error-prone, and delays trend identification.
Reactive Instead of Proactive Monitoring
Without real-time statistical analysis, process drift is detected only after batch failures. Late intervention increases rejects and regulatory risk.
Inconsistent Statistical Methods
Different analysts using different tools (Minitab, Excel) leads to inconsistent control chart methodologies and unreliable capability assessments.
No Automated Trend Detection
Manual reviews miss subtle process shifts. Nelson Rule violations and early capability deterioration go unnoticed until they cause quality events.
Disconnected from APQR Workflow
CPV data and APQR reporting operate in silos. No automated feed from continuous monitoring into annual product quality reviews.
Weak Linkage Between Process Stages
Limited traceability between Stage 1 (process design), Stage 2 (qualification) and Stage 3 (continued verification) creates gaps in validation lifecycle.
Resource-Intensive Reporting
Preparing CPV reports manually consumes 40-60 hours per product per quarter, diverting QA resources from proactive quality improvement.
Audit Readiness Gaps
Fragmented documentation and static snapshots make it difficult to demonstrate ongoing state of control during regulatory inspections.
FDA Process Validation Lifecycle: Three Stages of CPV Compliance
CPV (Stage 3) ensures the process remains in a validated state throughout commercial manufacturing
Stage 1
Process Design
Define critical process parameters, quality attributes, and design space based on scientific understanding.
- Define CPPs and CQAs
- Establish design space and proven acceptable ranges
- Risk assessment and control strategy
- Define monitoring plan for Stage 3
Stage 2
Process Qualification
Demonstrate that the process design can be reproduced consistently at commercial scale.
- Equipment and facility qualification (IQ/OQ/PQ)
- Process performance qualification (PPQ) batches
- Establish initial capability indices
- Confirm control strategy effectiveness
Stage 3
Continuous Process Verification
Ongoing assurance that the process remains in a state of control during routine commercial production.
- Continuous CPP/CQA monitoring and trending
- Statistical process control (SPC) charting
- Nelson Rule violation detection
- Capability index (Cp, Cpk, Pp, Ppk) tracking
- Automated alerts for process drift
- Feed into APQR for holistic quality review
CPV Software for Pharmaceutical Manufacturing
Continued process verification software with real-time process performance monitoring, automated statistical analysis, and inspection-ready CPV reporting
Real-Time Data Ingestion
Manufacturing process monitoring software with automated data pulls from LIMS, MES, ERP, and instruments. Single source of truth for all CPP and CQA data with validation and audit trail.
Automated SPC Charting
Control charts, trend lines, and capability analyses generated automatically for every monitored parameter without manual intervention.
Intelligent Alerting
Configurable alert thresholds for process drift, Nelson Rule violations, and capability deterioration with escalation workflows.
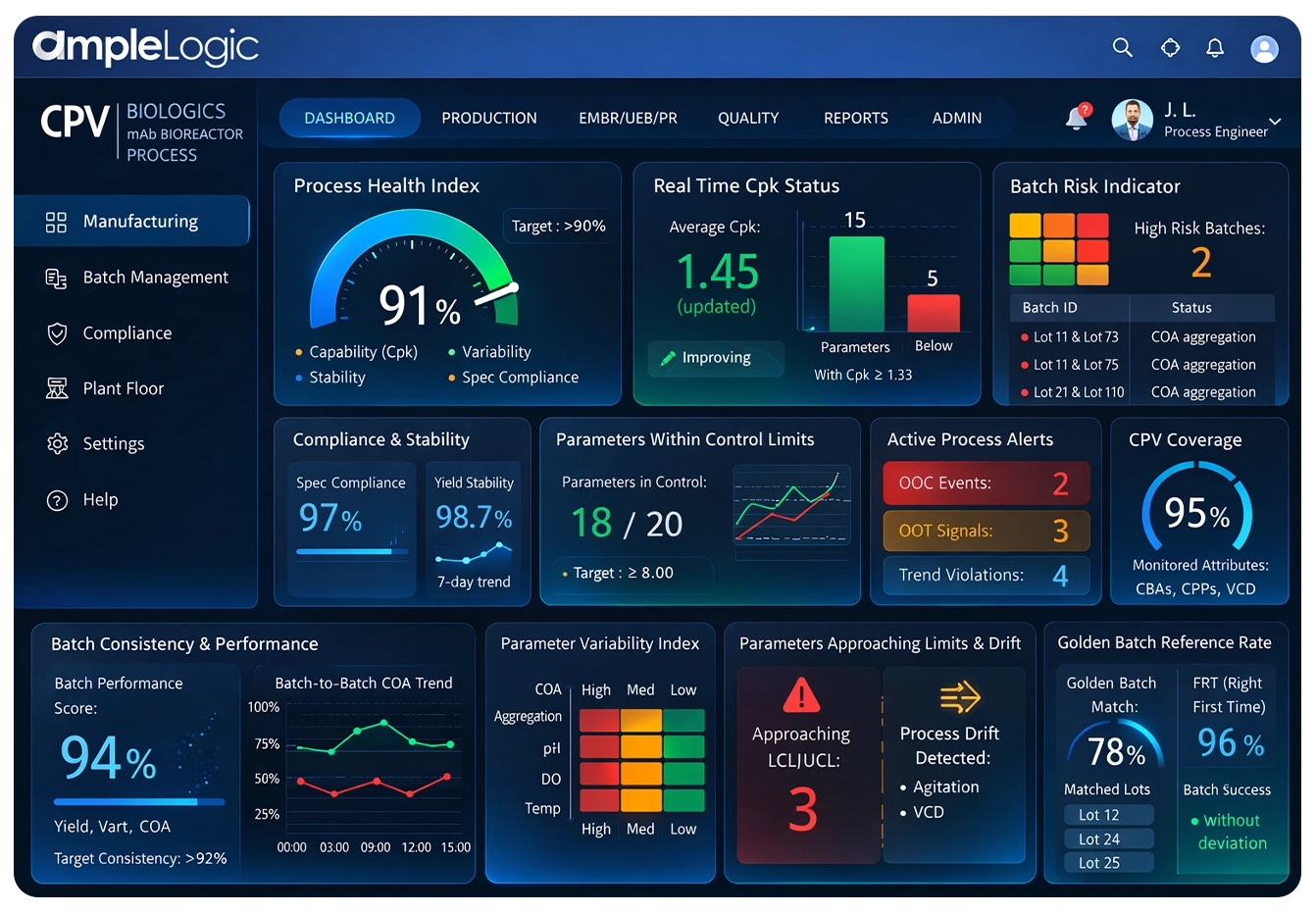
CPV Dashboard & Batch Process Monitoring
Centralized real-time batch process monitoring system showing process health across all products, parameters, and sites with drill-down capability.
Report Automation
Auto-generate CPV reports with embedded charts, statistical summaries, and conclusions. CPV software for pharmaceutical manufacturing with pre-formatted templates for regulatory submissions.
APQR Integration
Seamless data flow from CPV monitoring into APQR workflows. Stage 3 evidence automatically populates annual product quality reviews.
Real-Time CPP & CQA Monitoring for Continued Process Verification
Comprehensive monitoring of critical process parameters and quality attributes across every batch
Critical Process Parameters (CPPs)
- Temperature profiles and hold times
- Mixing speeds and durations
- Compression force and tablet hardness
- Drying endpoints and moisture content
- Granulation parameters and particle size
- Coating thickness and spray rates
Critical Quality Attributes (CQAs)
- Assay and potency values
- Content uniformity and blend uniformity
- Dissolution profiles and release rates
- Impurity levels and related substances
- pH, viscosity, and particle size distribution
- Sterility and endotoxin levels
Process Performance Indicators
- Batch yield and reconciliation
- In-process control results
- Cycle time and throughput
- Environmental monitoring data
- Equipment performance metrics
- Supply chain and raw material data
Batch-Wise Trending
Process performance monitoring software that generates trends batch-wise for CPPs, CQAs, manufacturing yield, and packing yield across all monitored parameters.
Automated Alert System
Configurable alerts for OOS, OOT, and process drift with automatic escalation to QA and production teams.
Golden Batch Benchmarking
Identify and benchmark golden batches for comparison, enabling best-in-class manufacturing performance tracking.
Multi-Product Monitoring
Monitor CPPs and CQAs across multiple products simultaneously with consolidated and individual views.
AmpleLogic CPV Statistical Process Control (SPC) Tools for GMP Manufacturing
Automated control charts, capability indices, and advanced statistical analysis for every CPP and CQA
Control Charts (I-MR, X-bar R)
Auto-generated I-chart, Moving Range, X-bar and R charts with control limits, specification limits, and centerlines for every CPP/CQA.
Capability Indices
Automated calculation of Cp, Cpk, Pp, Ppk, and process sigma. Track capability trends over time to detect early deterioration.
Six-Pack Reports
Comprehensive six-pack reports combining I-chart, moving range chart, histogram, normal probability plot, and capability analysis.
Nelson Rule Detection
Automated detection of all 8 Nelson Rule violations. Flagged violations trigger investigation workflows and corrective actions.
R-Based Statistical Engine
Built-in R statistical engine for advanced analyses including ANOVA, regression, multivariate analysis, and custom statistical tests.
Univariate & Multivariate Analysis
Support for both univariate and multivariate process analysis. Detect correlations between CPPs and CQAs for deeper process understanding.
AI-Driven Process Intelligence Platform for CPV
Predictive analytics, anomaly detection, and automated insights for proactive process control
Predictive Process Analytics
AI models analyze historical batch data to predict process drift before it impacts product quality. Early warning for capability deterioration.
Automated Trend Interpretation
AI-generated summaries interpret statistical trends, identify root causes, and recommend corrective actions for process deviations.
OCR Data Extraction
Extract data from scanned documents, handwritten logbooks, and legacy paper records. Automate data entry and ensure consistency.
AI ChatBot for Process Insights
Query historical process data, ask about trends, and get instant answers about process performance through natural language interaction.
Anomaly Detection
Machine learning algorithms detect unusual patterns in process data that traditional statistical methods may miss. Reduce false negatives.
Automated Report Narratives
AI generates section-wise summaries and conclusions for CPV reports, reducing manual narrative writing by 90%.
CPV Software Integrations with APQR, LIMS, eQMS, eBMR & Enterprise Applications
CPV data flows seamlessly between all quality and manufacturing systems
APQR
Direct feed of CPV data into APQR workflows and reports
eQMS
Deviations, CAPAs, OOS/OOT linked to CPV findings
LIMS
Analytical results, stability data, and lab incidents
eBMR
In-process data, batch records, and yield data
DMS
SOPs, protocols, and validation documents
eLOG
Equipment, area, and process logbook data
CAPS
Instrument calibration and equipment maintenance status
CVS
Cleaning verification and validation data
ERP
Raw material data and supply chain traceability
RIMS
Regulatory submission and compliance data
Benefits of Digital CPV Software for Pharmaceutical Quality Monitoring
Proactive quality control, faster reviews, and audit readiness through automated manufacturing process monitoring software
Proactive Quality Control
Detect process drift before it causes batch failures. Continuous process verification software for proactive manufacturing quality monitoring and management.
75% Effort Reduction
Automate data collection, chart generation, and report writing. Free QA teams to focus on investigations and improvements.
Real-Time Process Visibility
Live dashboards show process health across all products and sites. Drill-down from overview to individual parameter charts.
Eliminate Manual Minitab
Replace manual CSV exports and Minitab charting with automated, auditable, and always up-to-date statistical analysis.
Seamless APQR Integration
CPV findings automatically feed into annual product quality reviews. Consistent data and methodology across Stage 3 and APQR.
Faster Batch Release
Real-time monitoring and automated reviews enable faster batch disposition. Reduce review-to-release cycle time significantly.
Audit-Ready Documentation
Immutable audit trails, electronic signatures, and pre-formatted reports ensure inspection readiness at all times.
Multi-Site Harmonization
Standardize CPV methodologies and reporting across all manufacturing sites. Global consistency with local flexibility.
Multi-site CPV/OPV implementation with automated trending, statistical analysis, and seamless APQR integration. Harmonized global templates while supporting site-specific data models, workflows, and regulatory requirements.
Digital CPV implemented across manufacturing sites with automated statistical analysis, real-time dashboards, and integrated APQR reporting. Dynamic templates aligned to customer SOPs and regulatory expectations.
CPV Software Built for Global GxP Regulatory Compliance
Pre-built controls and documentation for FDA, EMA, MHRA, WHO, and ICH requirements
FDA Process Validation Guidance
Full compliance with FDA 2011 Process Validation Guidance covering Stage 3 Continued Process Verification requirements.
ICH Q8 / Q9 / Q10
Aligned with ICH guidelines for Pharmaceutical Development, Quality Risk Management, and Pharmaceutical Quality System.
EU GMP Annex 15
Compliance with EU requirements for qualification and validation including ongoing process verification.
21 CFR Part 11
Electronic records and signatures, audit trails, access controls, and data integrity assurance throughout.
MHRA Data Integrity
ALCOA+ principles enforced across all data collection, storage, and reporting activities.
WHO Technical Report Series
Aligned with WHO guidelines for process validation and continued process verification in pharmaceutical manufacturing.
Why Choose AmpleLogic CPV Software for Continued Process Verification
Unified APQR + CPV Platform
Lifecycle process validation software providing a single platform for both annual product quality review and continued process verification with shared data and analytics.
Compliant with Major Regulators
Pre-built controls and documentation for USFDA, EMA, MHRA, WHO, and ICH Q8-10 validation requirements.
10x Faster Deployments
Pre-validated templates and configurable workflows accelerate deployment from months to weeks.
200+ Professional Team
Dedicated resources for integration, validation, implementation, and ongoing product updates.
Continuous Product Upgrades
Regular updates with validation-ready change controls, new statistical methods, and enhanced AI capabilities.
Industries We Serve
Trusted across pharma, biotech, API manufacturers, CDMOs, and more
Pharmaceuticals
GMP-compliant process verification software for drug manufacturing with CPP/CQA trending and batch analysis
Biotechnology
Biologics and biosimilar CPV with complex analytical trending and process characterization
API Manufacturers
Active pharmaceutical ingredient CPV with process capability tracking and impurity trending
CDMOs
Multi-client CPV management with product-specific monitoring and reporting templates
Biosciences
QC Lab continuous process verification software with advanced multivariate analysis and process optimization for R&D and commercial manufacturing
CROs
Contract research CPV with multi-client data management and analytical trending
Medical Devices
Device manufacturing CPV with performance trending and design control integration
Food & Beverages
Food processing CPV with safety parameter monitoring and HACCP compliance
Ready for Proactive CPV Process Verification Software?
AmpleLogic CPV monitoring software transforms manufacturing quality monitoring from reactive to proactive. Automated CPP/CQA monitoring, real-time statistical analysis, and audit-ready documentation for continuous process verification.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.