제약 분야를 위한 전자 문서 관리 시스템(EDMS)
AmpleLogic의 제약 문서 관리 소프트웨어는 제약 등급 컴플라이언스, 바코딩, 관리된 SOP 관리, 인쇄 발급, 버전 및 대체 관리, 지능형 자동화를 갖춘 엔터프라이즈급 문서 관리 소프트웨어를 제공합니다.
25%
감사 지적 사항 감소
30%
출시 시간 개선
50%
수동 작업 절감
FDA
21 CFR Part 11 준비 완료
제약 문서 관리 소프트웨어 없이 발생하는 주요 GMP 과제
제약 문서 관리에서 컴플라이언스, 효율성 및 추적성에 영향을 미치는 중요한 문제점
종이 기반 또는 부분적으로 디지털화된 제약 문서 시스템이 여전히 존재
컴플라이언스 문서 관리 시스템 없이 수동 문서 추적은 주당 15-20시간을 소비하여 오류 및 검색 시간이 증가합니다.
EDMS 기능 격차: 취약한 SOP 버전 관리 및 제한된 인쇄 발급
문서 버전 관리 소프트웨어 부재로 운영자가 오래된 SOP를 사용하게 되어 감사 지적 사항 및 컴플라이언스 위반으로 이어집니다.
바코드 기반 문서 추적 없음 및 취약한 대체 관리
관리된 사본 추적 또는 자동화된 폐기 없이 구식 문서가 계속 유통됩니다.
LMS, eQMS, LIMS 및 MES 시스템과의 DMS 통합 어려움
연결되지 않은 시스템은 추적성을 저해하여 감사 준비 노력과 대응 시간을 증가시킵니다.
GMP 컴플라이언스를 위해 제약 등급 EDMS가 제공해야 할 것
문서 수명주기 관리, 규제 컴플라이언스, 관리된 SOP 관리 및 검증된 자동화
문서 수명주기 관리
작성부터 검토, 승인, 출시, 보관까지 관리된 대체 및 폐기를 포함하는 완전한 문서 수명주기 관리 소프트웨어.
검증 및 관리
규제 요건 및 디지털 문서 관리 시스템 기능에 매핑된 관리를 갖춘 검증된 문서 승인 워크플로 소프트웨어.
통합 및 자동화
수동 조정을 줄이기 위한 원활한 시스템 통합, 고급 검색 및 AI 기반 자동화.
21 CFR Part 11 및 EU Annex 11 규제 컴플라이언스
매핑된 관리를 갖춘 FDA, EU Annex 11 및 GxP 문서 관리 시스템 요건을 충족하는 21 CFR Part 11 준수 문서 관리 시스템.
추적성 및 보안
강력한 문서 버전 관리 소프트웨어, 감사 추적, 전자 서명 및 관리된 사본 인쇄 및 검색 추적.
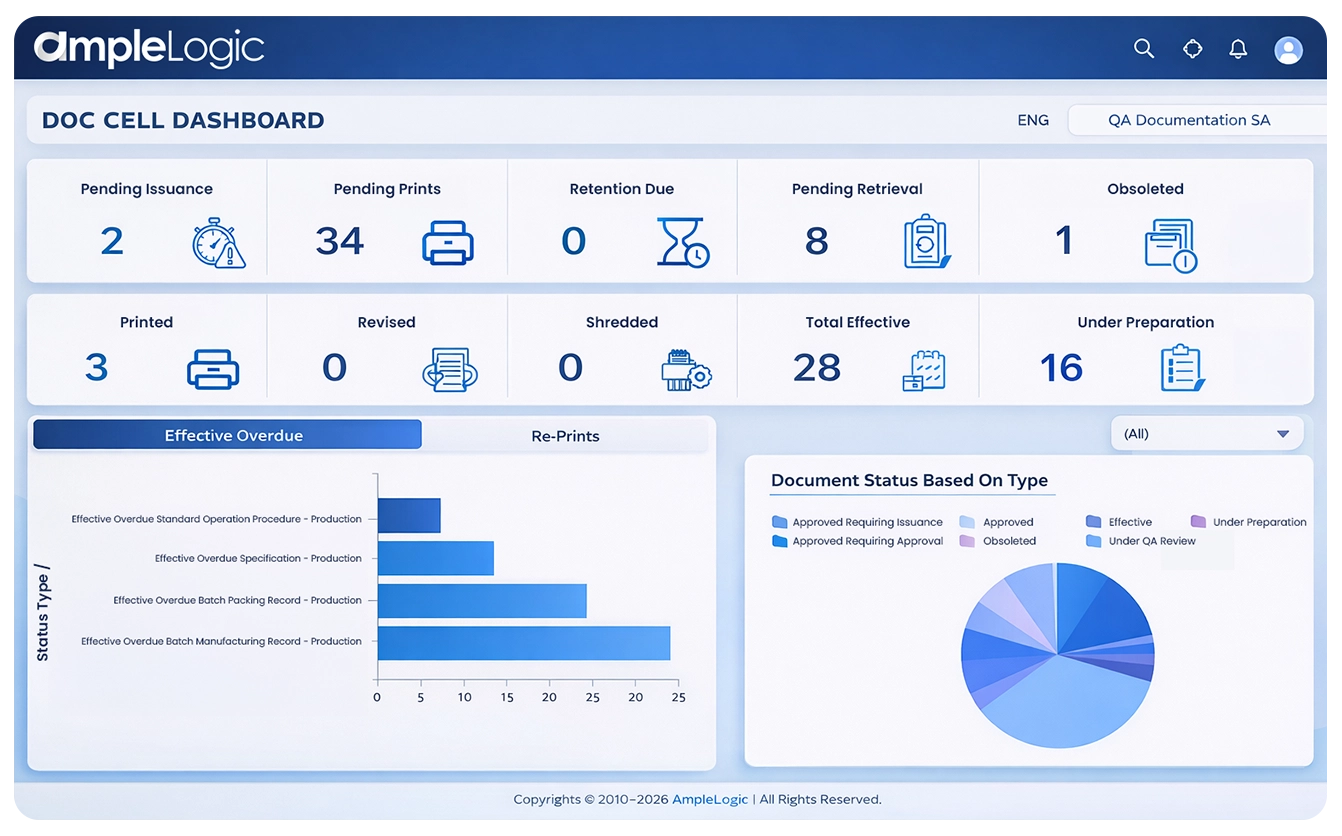
제약 문서 관리 소프트웨어에서 관리되는 핵심 GMP 문서 유형
GMP 준비를 위한 감사 추적 및 메타데이터를 갖춘 문서 수명주기 관리 소프트웨어
품질 문서
- 버전 관리가 포함된 표준 운영 절차(SOP)
- 승인 워크플로가 있는 변경 관리 기록
- 컴플라이언스 추적성을 위한 연결된 교육 기록
- 폐기된 사본 관리 및 대체 관리
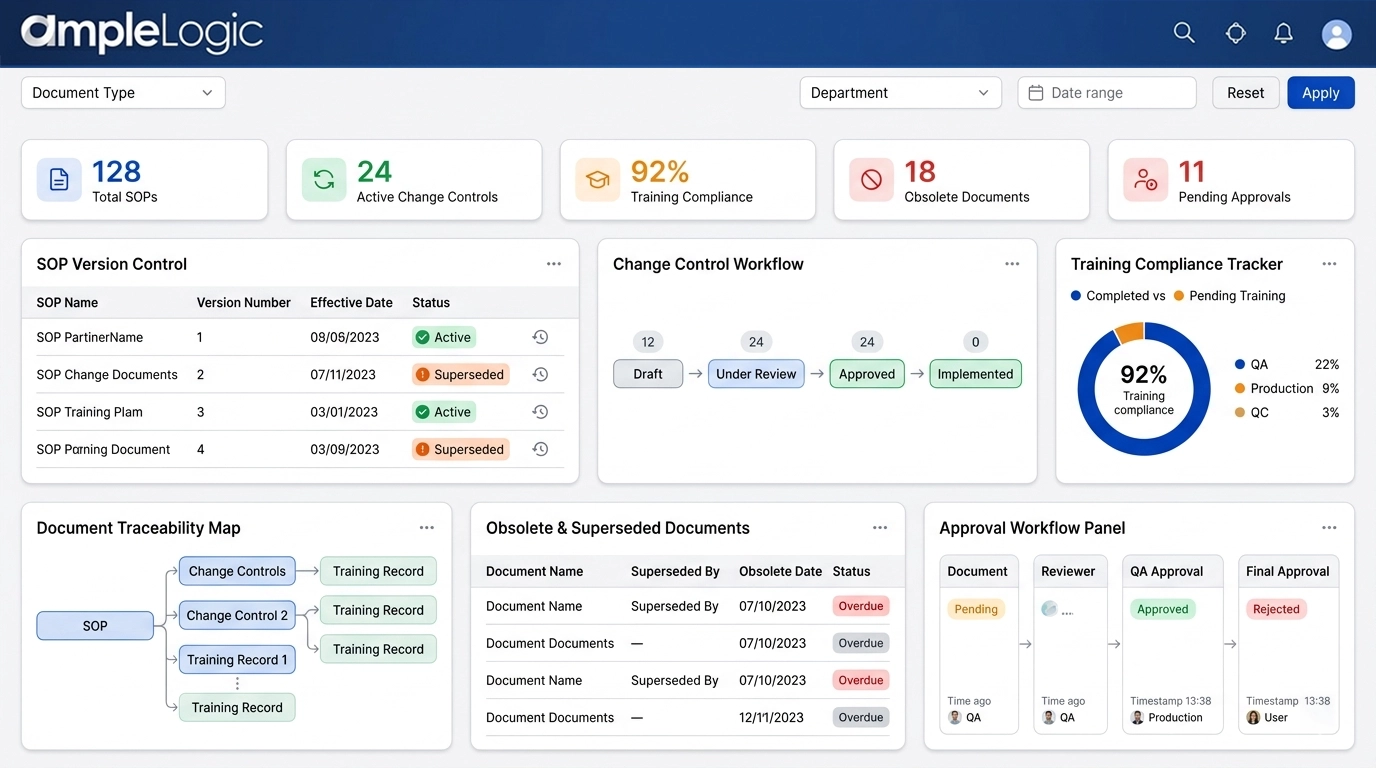
SOP 버전 관리 소프트웨어: 대체, 구식 사본 및 수명주기 관리
디지털 문서 관리로 현재 문서만 규제 프로세스를 구동하도록 보장
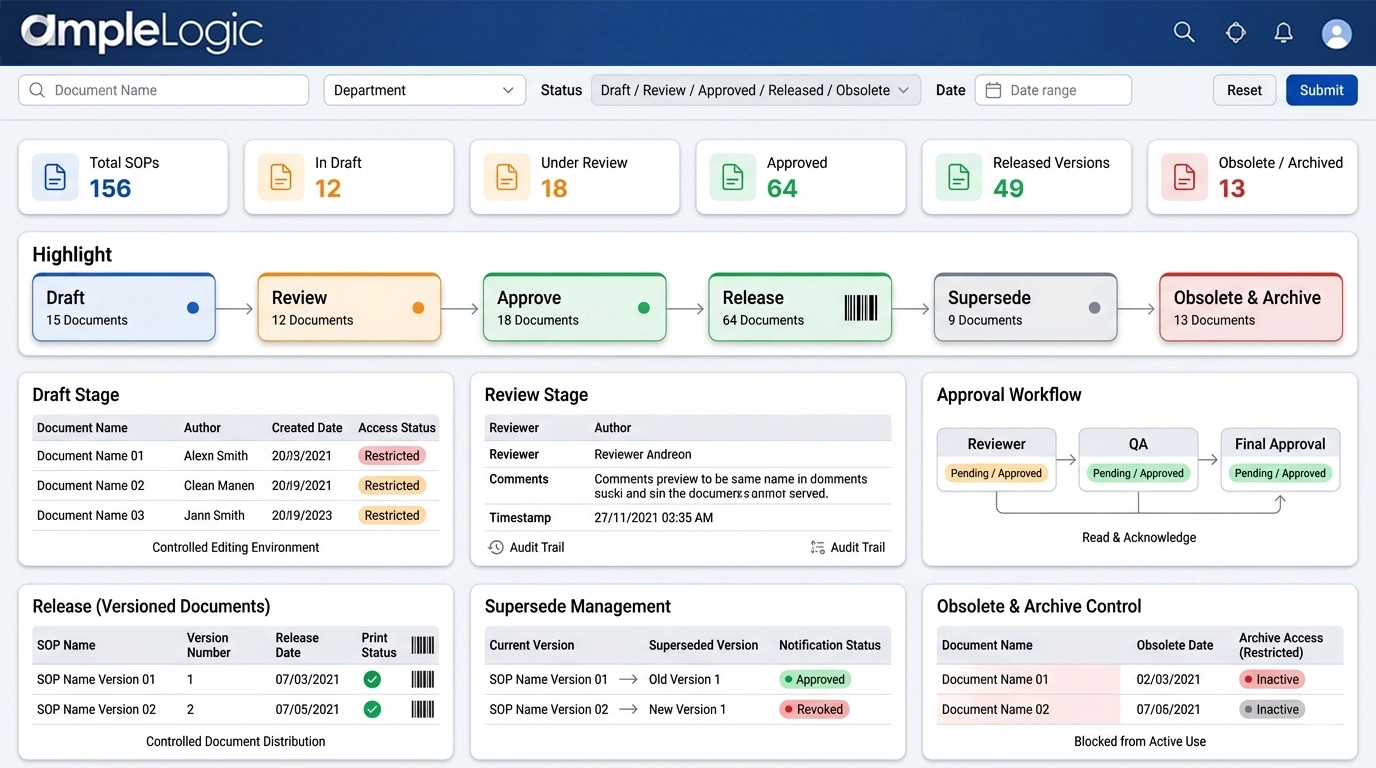
초안
명확한 초안 상태와 제한된 접근으로 관리된 문서 편집.
검토
날짜/시간 스탬프가 있는 검토자 의견의 완전한 감사 추적과 함께 교차 기능 팀에 의한 병렬 검토.
승인
읽기 및 확인 단계가 강제 적용되는 공식 승인 워크플로.
출시(버전화)
인쇄본에 대한 버전 번호 부여, 인쇄 관리 및 바코드 할당.
대체
이전 버전의 자동화된 대체 및 사용자에 대한 알림.
폐기 및 보관
구식 문서의 명시적 철회, 관리된 보관 접근 및 활성 프로세스에서의 사용 차단.
초안
명확한 초안 상태와 제한된 접근으로 관리된 문서 편집.
검토
날짜/시간 스탬프가 있는 검토자 의견의 완전한 감사 추적과 함께 교차 기능 팀에 의한 병렬 검토.
승인
읽기 및 확인 단계가 강제 적용되는 공식 승인 워크플로.
출시(버전화)
인쇄본에 대한 버전 번호 부여, 인쇄 관리 및 바코드 할당.
대체
이전 버전의 자동화된 대체 및 사용자에 대한 알림.
폐기 및 보관
구식 문서의 명시적 철회, 관리된 보관 접근 및 활성 프로세스에서의 사용 차단.
인쇄 발급, 관리된 사본 배포 및 검색 추적
비관리, 교육 및 초안 사본을 포함하여 현재의 승인된 문서만 생산에 전달되도록 보장
승인
문서 승인 워크플로 소프트웨어를 통한 버전 및 승인 메타데이터로 문서 완료.
버전 잠금
승인된 버전 동결; 디지털 문서 관리 시스템 보호 장치를 사용하여 편집 방지 및 대체된 상태 표시.
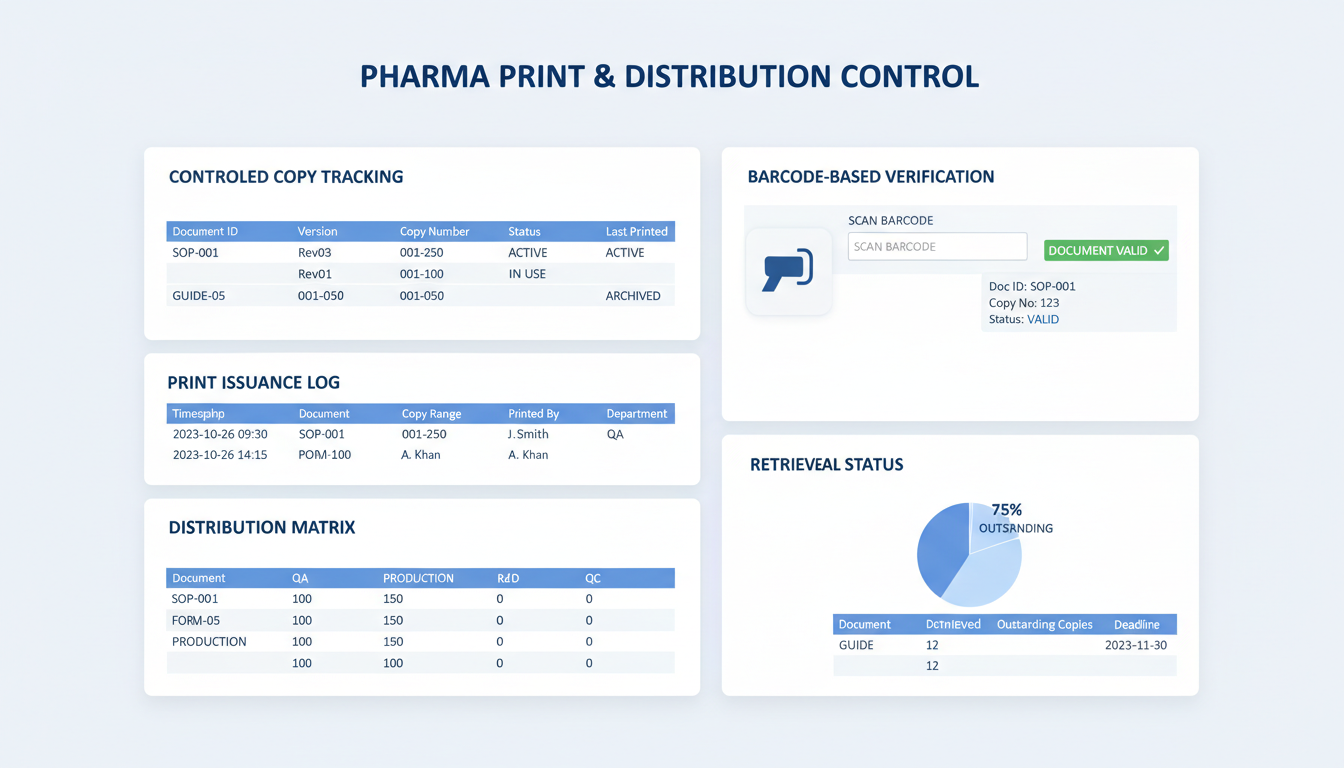
인쇄 발급
역할/사이트별 인쇄 권한 부여, 관리된 사본, 비관리 사본, 교육 사본 및 초안 사본과 같은 워터마크 생성. 사이트 전반에 걸쳐 관리된 사본 인쇄 및 검색 활성화.
배포
바코드 레이블 및 인수 확인과 함께 관리된 사이트 및 역할 기반 목록에 게시.
조정 및 감사 추적
완전한 제약 문서 관리 시스템 추적성을 위해 인쇄 세부 사항을 추적하고 감사와 함께 발급된 종이 사본을 조정.
인쇄 관리 및 검증
바코드 기반 추적을 통한 디지털 시스템과 현장 실물 사본 간의 격차 해소
동적 바코드 생성
모든 인쇄된 실물 사본에 대한 자동 워터마킹 및 고유 바코드 생성.
사용 시점 검증
문서 승인 상태 및 버전 확인을 위한 생산 현장에서의 실시간 바코드 스캔.
종합적인 인쇄 감사
사용자별 인쇄 로그 및 위치 데이터를 포함한 배포된 실물 사본의 완전한 추적.
GxP 컴플라이언스 문서 관리 시스템을 위한 지능형 OCR 및 AI 검색
규제 문서를 디지털 문서 관리 시스템 전반에서 검색 가능하고 검토 가능하며 접근 가능하게 만들기
AI 기반 OCR
스캔 및 이미지를 검색 가능한 편집 가능 텍스트로 변환.
수동 전사를 줄이고 빠른 검색 가능.
지능형 검색
문맥적 관련성으로 규제 콘텐츠를 표면화.
문서 저장소 전반에 걸쳐 검색 정확도 향상.
음성 활성화 검색
핸즈프리 검색 및 검토 가속화.
감사 및 팀 워크플로 중 빠른 접근.
전자 발견 및 감사 지원
더 빠른 문서 검토, 증거 검색 및 감사 대응 가능.
컴플라이언스, 추적성 및 규제 감사 지원.
팀 전체 접근성
기능 전반에 걸쳐 관리된 문서에 대한 일관된 접근 제공.
규제 환경에서 추적성 및 협업 가능.
eQMS, LMS, LIMS 및 MES와의 GMP 준수 DMS 통합
엔드투엔드 문서 추적성을 위해 엔터프라이즈 GxP 문서 관리 시스템 생태계에 원활하게 연결
LMS
새로운 SOP 도입, 버전 변경으로 인한 교육 필요사항. 교육 완료 확인.
eQMS
변경 관리, 감사 지적 사항, CAPA에 의한 버전 변경.
RIMS
규제 지적 사항으로 인한 버전 변경.
LIMS
실험실 결과 및 기록과 함께 시험 방법 및 STP.
전자 로그북
로그북 템플릿 참조.
세척 밸리데이션
세척 절차를 위한 SOP 접근.
eBMR/MES
BMR/BPR 문서 관리 및 접근.
CAPS
교정 절차를 위한 장비 매뉴얼/SOP.
SAP
전사적 자원 관리 통합.
UMS
사용자 관리 시스템 통합.
AmpleLogic 로우코드 DMS 플랫폼 강점 및 AI 기반 가치
생명과학을 위한 로우코드 유연성, 컴플라이언스 준비 및 관리된 자동화
LCNC 드래그 앤 드롭 디자이너
시각적 빌더가 커스텀 코드를 줄이고 빠른 앱 구성을 위한 제공 속도를 높입니다.
제약을 위한 클라우드 문서 관리
가속화된 일정과 빠른 비즈니스 성과를 갖춘 제약 분야용 클라우드 문서 관리 소프트웨어 — 최소 3개월 내 ROI 실현.
규제 산업 문서 관리
문서화 및 감사를 위해 설계된 규제 산업 문서 관리 소프트웨어. USFDA, MHRA, ISO 준비 완료.
18개 이상의 COTS 애플리케이션
일반적인 생명과학 워크플로를 위한 즉시 배포 가능한 사전 구축 앱.
100개 이상의 생명과학 고객
고객 기반은 강력한 업계 견인력과 검증된 플랫폼 채택을 보여줍니다.
AI 기반 자동화
AI가 구성 및 처리 가속화를 위한 자동화, 추출 및 앱 조립을 지원합니다.
DMS 소프트웨어 ROI: 디지털 제약 문서 관리의 비즈니스 이점
컴플라이언스, 속도, 정확성 및 확장 가능한 디지털 품질
25%
감사 지적 사항 감소
배포 후 지적 사항의 목표 감소율.
30%
출시 시간 개선
문서 출시 시간의 예상 감소율.
50%
수동 작업 절감
수동 처리 시간의 예상 감소율.
샘플 SOP 및 배치 기록으로 라이브 DMS 데모 예약
AmpleLogic의 AI 기반 전자 문서 관리 시스템으로 관리된 인쇄, 인쇄 발급 및 엔드투엔드 추적성을 경험하십시오.
생명과학 분야의 최신 정보를 받아보세요
최신 제품 업데이트, 규정 준수 뉴스, 업계 인사이트를 받은 편지함으로 직접 받아보세요.