40%
材料廃棄の削減
100%
試薬トレーサビリティ
Zero
期限切れ材料の使用ゼロ
60%
調達サイクルの高速化
Industry Challenges
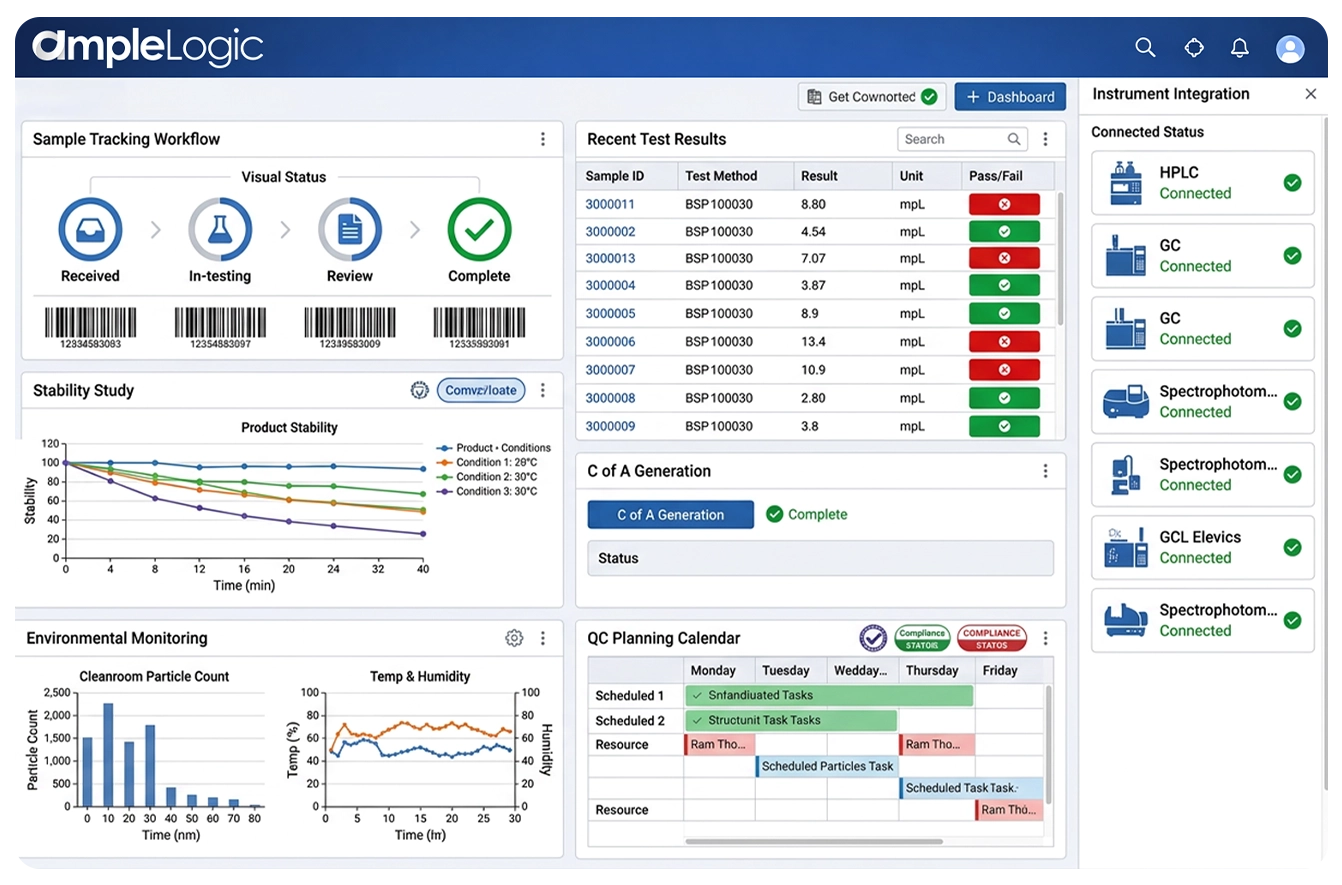
Why Laboratory Inventory Management Software for Pharmaceutical & Life Sciences QC Labs Matters
医薬品およびライフサイエンスラボラトリーは、数千種類の試薬、化学品、消耗品、標準品、HPLCカラムを管理しています。専用のラボラトリー在庫追跡ソフトウェアがなければ、ラボはスプレッドシートや手動ログブックに依存することになり — 試験での期限切れ材料の使用、在庫切れ、規制非準拠、予算の無駄遣いを引き起こします。AmpleLogicのラボラトリー在庫管理ソフトウェアは、LIMSに組み込まれた完全統合型ラボ材料管理システムによりこれらのリスクを排除します。
Common Challenges
スプレッドシートベースの手動追跡は試薬在庫管理および化学品在庫管理においてエラーを引き起こし、期限切れまたは規格外材料の使用リスクをもたらします
ラボラトリー在庫管理システムの欠如は在庫切れ、緊急調達、計画外のラボダウンタイムを招きます
集中型のラボラトリー消耗品追跡がないと重複注文、過剰購入、予算の非効率な活用が生じます
HPLCカラム使用ソフトウェアがないと、カラムの廃棄決定が推測に頼り、分析失敗や再試験を引き起こします
分断されたサンプル在庫追跡および安定性サンプル在庫追跡は、監査準備を時間がかかりエラーが発生しやすいものにします
QCラボラトリー在庫ソフトウェアがないと、複数拠点のラボは施設間の在庫レベルに統一された可視性を持てません
How the Platform Helps
すべての材料タイプおよびラボラトリー拠点にわたるリアルタイム在庫追跡を持つ統合LIMS在庫管理システムを提供します
ロットレベルのトレーサビリティと有効期限の強制により試薬在庫管理、製薬試薬管理、化学品在庫管理を自動化します
自動再注文ポイントアラートを備えた専用のラボラトリー消耗品追跡とラボ供給管理ソフトウェアを提供します
注入回数監視、性能トレンド分析、ルールベースのカラム廃棄のためのHPLCカラム使用ソフトウェアを統合します
FDA、MHRA、EMA検査に向けた完全な監査証跡を備えたサンプル在庫追跡および安定性サンプル在庫追跡をサポートします
企業規模の在庫管理のための包括的な製薬ラボ在庫管理およびQCラボラトリー在庫ソフトウェアプラットフォームとして機能します
Platform Capabilities
Core Capabilities
Integrated capability areas that power laboratory inventory management software for pharmaceutical & life sciences qc labs.
試薬・化学品ライフサイクル管理
受入から廃棄まで、ロット追跡、CoA取込、SDS統合、危険物分類、製薬試薬管理ワークフローを備えた完全な試薬在庫管理および化学品在庫管理
- ベンダー資格を含むロットレベルの試薬・化学品追跡
- 有効期限と再試験日の自動強制
- 安全コンプライアンスのためのSDSと危険物クラス統合
- 材料マスターレコードとCoA取込・保存
消耗品・ラボ供給追跡
フィルター、バイアル、ピペットチップ、ガラス器具、使い捨て品のための設定可能な安全在庫アラートと最小/最大再注文レベルを備えた自動ラボラトリー消耗品追跡とラボ供給管理ソフトウェア
- 計量単位換算を含むカテゴリーベースの消耗品カタログ
- 自動再注文ポイントアラートと購買依頼
- テスト、プロジェクト、またはコストセンター別の消耗分析
- サプライヤーパフォーマンススコアリングとリードタイム追跡
HPLC・GCカラム管理
分析失敗を防ぐため、HPLCおよびGCカラムの注入回数、バックプレッシャートレンド、プレートカウント性能、廃棄ルールを追跡する専用HPLCカラム使用ソフトウェア
- 機器統合に連携した注入回数追跡
- 性能劣化トレンド分析
- ルールベースのカラム廃棄と交換アラート
- 予算最適化のための注入当たりカラムコスト分析
サンプル・安定性在庫追跡
監査対応のための完全なトレーサビリティを備えた特定サンプル、バッチ、ICH安定性プログラムに対する材料消耗を記録する統合サンプル在庫追跡および安定性サンプル在庫追跡
- 完全な消耗トレーサビリティのための材料とサンプルの連携
- 安定性プルスケジュール連携サンプル在庫管理
- コスト配分のためのバッチレベル材料使用レポート
- すべての在庫トランザクションの監査証跡ログ
リアルタイム在庫可視化と管理
すべての拠点にわたる在庫レベル、保管場所、消耗パターン、調達状況への即時可視化を提供するリアルタイム在庫追跡ダッシュボードとラボラトリー在庫管理システム
- 複数拠点・複数倉庫の在庫レベルダッシュボード
- バーコードおよびRFID対応の受入、払出、返却
- 環境モニタリングを備えたジオマップ型保管場所
- 在庫回転率と廃棄率のための設定可能なKPIダッシュボード
調達・ベンダー統合
SAP、Oracle、NetSuiteと統合した、自動生成購買依頼から入荷検収、ベンダー評価、請求書照合までをカバーするQCラボラトリー在庫ソフトウェア内のエンドツーエンド調達自動化
- 安全在庫トリガーによる購買依頼の自動化
- ベンダー資格と承認済みサプライヤー管理
- バーコード/RFIDスキャンによる入荷検収確認
- シームレスな財務照合のためのERP統合
Feature Highlights
End-to-End Process Flow
How It Works
Structured workflow from initiation through closure with built-in compliance checkpoints and full traceability at every stage.
材料受入・登録
バーコード/RFIDスキャン、CoA取込、ベンダー情報を使用して試薬、化学品、消耗品、標準品をLIMS在庫管理システムに受入・登録します
保管割り当て・場所追跡
ラボ材料追跡システムを通じて環境条件モニタリングと棚マッピングにより、材料を指定保管場所(冷蔵庫、冷凍庫、化学品キャビネット)に割り当てます
払出・消耗追跡
スキャンベースの確認により特定サンプル、テスト、バッチに対して材料を払出します。サンプル在庫追跡を通じて消耗量を追跡し、完全な使用トレーサビリティを維持します
有効期限・再試験日監視
期限切れや劣化した材料の使用を防ぐための有効期限、再試験日、保存期間上限に対する自動アラート — ラボラトリー在庫追跡ソフトウェアの主要機能
再注文・調達自動化
ラボ供給管理ソフトウェアによる在庫が安全在庫レベルを下回った際のベンダー管理と承認ワークフローによる購買依頼の自動トリガー
在庫レポートとコンプライアンス監査
ラボラトリー在庫管理ソフトウェアからGMP/GLP検査向けのリアルタイム在庫レポート、消耗トレンド分析、廃棄追跡、監査対応ドキュメントを生成します
Capabilities
Key Features
Comprehensive capabilities designed for regulated laboratory environments and operational excellence.
試薬在庫管理
ロットレベル追跡、有効期限、分析証明書、使用履歴を備えた包括的な試薬管理
化学品在庫管理
SDS統合、危険分類、安全在庫アラートを含むエンドツーエンドの化学品在庫管理
ラボ消耗品追跡
フィルター、バイアル、ピペットチップ、ガラス器具、ラボ用品の再発注点アラート付き自動追跡
HPLCカラム使用ソフトウェア
HPLCおよびGCカラムの注入回数、パフォーマンス履歴、廃棄基準の追跡
保管場所・棚マッピング
冷蔵庫、冷凍庫、薬品棚を含む保管場所のバーコード/RFID追跡によるマッピングと管理
有効期限・有効期間モニタリング
有効期限、再試験日、有効期間の接近に対する自動アラート
サンプル在庫追跡
完全なトレーサビリティのために特定のサンプル、テスト、バッチに対する資材消費を記録
自動再発注・調達
在庫が安全在庫レベルを下回った際の購入要求の自動トリガー
標準品管理
認証追跡と保管条件を含む一次および作業用標準品の管理
在庫レポート・ダッシュボード
在庫レベル、消費トレンド、廃棄分析、コンプライアンスレポートのリアルタイムダッシュボード
Value Delivered
QCラボ在庫管理 - 主なメリット
Measurable outcomes that transform laboratory operations and deliver enterprise-wide value.
期限切れ材料リスクの排除
有効期限と再試験日の自動監視により期限切れ試薬・化学品の払出をブロックし、準拠した材料のみが試験に使用されることを保証します — ファーマラボラトリー在庫管理の重要な機能
完全な規制トレーサビリティ
ラボ材料追跡システムは材料受入から特定サンプルおよびバッチへの消耗まで、FDA 21 CFR Part 11、EU Annex 11、ISO 17025要件を満たすエンドツーエンドの監査証跡を維持します
調達コストの削減
スマートな再注文ポイント自動化と消耗分析を備えたラボ供給管理ソフトウェアは緊急注文を削減し、過剰在庫を最小化し、ラボ拠点全体の予算配分を最適化します
カラム・機器ROIの最大化
HPLCカラム使用ソフトウェアはデータ駆動の廃棄決定によりカラム寿命を延ばし、分析コストを削減し、劣化したカラムによる分析失敗を防止します
企業規模の在庫可視化
ラボラトリー在庫管理ソフトウェアは複数拠点の運営全体でリアルタイム在庫追跡を提供し、マネージャーが在庫をバランスさせ、調達を集約し、拠点レベルの在庫切れを防止できます
監査対応の加速
組み込みの監査証跡、電子署名、コンプライアンスレポートを備えたQCラボラトリー在庫ソフトウェアにより、ラボがFDA、MHRA、EMA、WHO監査に常に検査対応できることを保証します
Real-World Applications
Use Cases
Proven scenarios where this module delivers measurable value across laboratory and manufacturing operations.
製薬QCラボ -- 試薬・化学品管理
QCラボラトリー在庫ソフトウェアは自動有効期限アラート、ロットトレーサビリティ、規制監査時の製薬試薬管理のためのCoA管理により複数のQCラボ全体で数百種類の試薬と化学品を追跡します
試験でゼロの期限切れ試薬使用、FDA検査向け100%ロットトレーサビリティ
HPLC/GCラボラトリー -- カラムライフサイクル最適化
HPLCカラム使用ソフトウェアは製薬ラボ在庫管理における分析精度を確保するためにカラムライフサイクルを最適化し、カラムの注入回数、性能劣化、廃棄基準を監視します
最適化された使用追跡で30%長いカラム寿命、分析失敗の削減
複数拠点運営 -- 集中型在庫管理
ラボラトリー在庫管理システムは在庫レベル、消耗パターン、調達状況の集中型リアルタイム在庫追跡により複数のラボラトリー拠点にわたる在庫管理を標準化します
統一された在庫可視化、緊急調達注文40%削減
安定性ラボ -- 安定性サンプル在庫追跡
安定性サンプル在庫追跡はサンプルプルスケジュールを材料消耗に連携させ、すべての安定性時点に試薬と消耗品が利用可能であり、使用されたすべての材料が規格内であることを保証します
材料不足による安定性プルの見落としゼロ、完全なICH監査証跡
Common Questions
Frequently Asked Questions
Ready to Modernize 医薬品・ライフサイエンスQCラボ向けラボラトリー在庫管理ソフトウェア?
See how AmpleLogic LIMS can transform your 医薬品・ライフサイエンスqcラボ向けラボラトリー在庫管理ソフトウェア workflows with a personalized demo.
ライフサイエンスの最前線へ
最新の製品アップデート、コンプライアンスニュース、業界インサイトをお届けします。