高度安定性管理
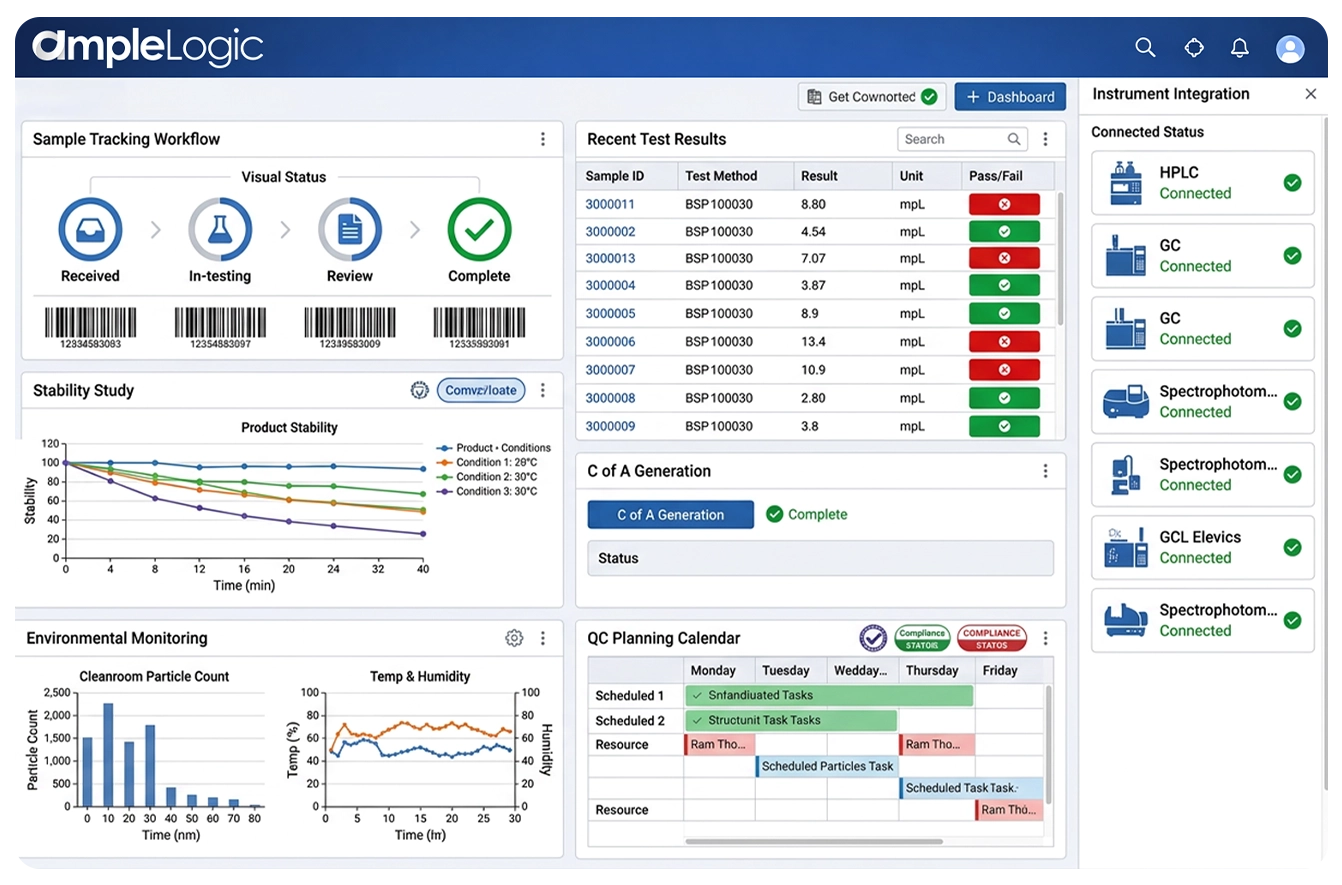
AmpleLogic is an enterprise stability study management software and stability schedules management system that delivers end-to-end stability program management — from stability protocol creation and sample pull scheduling through chamber storage management, test execution, stability trend analysis, shelf-life determination, and regulatory-ready reporting aligned to ICH Q1A stability guidelines, FDA stability requirements, and GMP stability programs.
40%
Faster Shelf-Life Determination
100%
ICH Q1A Guideline Compliance
50%
Less Manual Analyst Effort
3x
Concurrent Study Capacity
Industry Challenges
Why Advanced Stability Management Matters
AmpleLogic Advanced Stability Management is a comprehensive stability study management software that unifies the complete stability study lifecycle — stability protocol creation, sample pull scheduling, chamber storage management, test execution, trend analysis and reporting, and shelf-life determination — in a single stability testing management system with built-in ICH Q1A stability guidelines compliance, FDA stability requirements alignment, and GMP stability program support.
Common Challenges
Manual stability protocol creation leads to inconsistent study designs and regulatory gaps
Spreadsheet-based sample pull scheduling causes missed pull points and data integrity risks
Fragmented chamber storage management limits visibility into environmental conditions and excursions
Disconnected test execution workflows create traceability gaps between stability studies and results
Manual trend analysis and reporting delays shelf-life determination and regulatory submissions
Lack of integrated expiry determination analytics increases risk of inaccurate product shelf-life evaluation
How the Platform Helps
Automate stability protocol creation with ICH Q1A-compliant templates and configurable study designs
Eliminate missed pull points with automated sample pull scheduling and calendar-based reminders
Centralize chamber storage management with real-time monitoring and excursion alerts
Streamline test execution with integrated LIMS workflows and direct instrument connectivity
Accelerate shelf-life determination and expiry determination with AI-powered trend analysis
Ensure regulatory compliance with FDA stability requirements and GMP stability programs
Platform Capabilities
Core Capabilities
Integrated capability areas that power advanced stability management.
Stability Study Lifecycle Management
Manage the complete stability study lifecycle from study initiation through protocol creation, sample management, testing, trending, and closure in a unified stability study management software.
- ICH Q1A-compliant stability protocol creation
- Automated sample pull scheduling with reminders
- Chamber storage management and environmental monitoring
- Integrated test execution with LIMS
- Stability trend analysis and statistical reporting
- Study closure with regulatory-ready documentation
Shelf-Life Determination & Expiry Management
AI-powered product shelf-life evaluation and expiry determination with statistical trend analysis, degradation curve fitting, and confidence interval-based shelf-life projection.
- Statistical shelf-life projection with confidence intervals
- Product shelf-life evaluation against acceptance criteria
- Expiry determination and retest date calculation
- Stability trend analysis with degradation modeling
- Accelerated and long-term stability correlation
- Label claim support with statistical evidence
Regulatory Compliance Engine
Built-in compliance with ICH Q1A stability guidelines, FDA stability requirements, GMP stability programs, and global regulatory frameworks for stability testing management.
- ICH Q1A-Q1F guideline alignment and validation
- FDA 21 CFR Part 211.166 stability requirements
- GMP stability program management and ongoing commitments
- EU Annex 11 computerized system compliance
- MHRA and global health authority readiness
- Audit-ready documentation and submission reports
AI-Driven Stability Intelligence
Machine learning models analyze historical stability data, degradation patterns, and environmental factors to deliver predictive insights for accelerated stability program management.
- Predictive shelf-life forecasting
- Early degradation risk detection
- Anomaly detection in stability results
- Cross-study trend pattern recognition
- Automated statistical analysis and charting
- Decision support for retest and expiry evaluation
Environmental Chamber Control
Comprehensive chamber storage management with real-time sensor integration, environmental condition monitoring, excursion management, and chamber capacity planning.
- Real-time temperature and humidity monitoring
- Chamber capacity and allocation planning
- Excursion detection with automated alerts
- Chamber qualification and maintenance tracking
- Multi-zone environmental condition mapping
- Historical environmental data trending
Feature Highlights
End-to-End Process Flow
How It Works
Structured workflow from initiation through closure with built-in compliance checkpoints and full traceability at every stage.
Stability Protocol Creation
Define ICH Q1A-compliant stability protocols with automated study designs, sampling schedules, storage conditions, acceptance criteria, and configurable testing time points
Stability Sample Scheduling
Automated sample pull scheduling with calendar-based reminders, dynamic rescheduling, priority-based pull management, and complete audit trails for every pull event
Stability Chamber and Environmental Monitoring
Monitor stability storage conditions such as temperature and humidity across stability chambers and warehouses. Tracks environmental conditions, triggers alerts for excursions, and links stability data with sample results
Test Execution
Execute analytical tests within the integrated LIMS workflow with direct instrument connectivity, in-line calculations, and full sample-to-result traceability
Sample Trend Analysis & Shelf-Life Determination
AI-powered stability trend analysis with degradation modeling, statistical shelf-life projection, product shelf-life evaluation, expiry determination, and confidence interval reporting
Regulatory Reporting & Submission
Generate ICH-compliant stability reports, FDA submission packages, and GMP stability program documentation with complete trend data, statistical summaries, and deviation tracking
Capabilities
Key Features
Comprehensive capabilities designed for regulated laboratory environments and operational excellence.
安定性プロトコル作成
設定可能な試験デザイン、保管条件、許容基準、自動サンプリングスケジュールを備えたICH Q1A準拠安定性プロトコルの定義
安定性サンプルスケジューリング
カレンダーベースのリマインダー、動的再スケジューリング、完全な監査証跡を備えた自動サンプルプルスケジューリング
チャンバー・環境モニタリング
安定性チャンバーおよび倉庫全体の温度・湿度などの安定性保管条件のモニタリング
試験実行・データ取込
機器直接統合と完全なトレーサビリティを備えた同一LIMSワークフロー内での分析試験の実行
安定性トレンド分析・報告
劣化曲線フィッティング、管理図、規制対応安定性レポートを含む自動統計トレンド分析
AI搭載有効期限決定
信頼区間、早期有効期限リスクアラート、加速安定性予測を備えた機械学習駆動の有効期限予測
有効期限決定・再試験日管理
安定性トレンドデータに基づく体系的な有効期限決定、再試験日計算、自動ラベル表示サポート
ICH Q1A規制コンプライアンス
製薬・バイオテクノロジーの安定性プログラム向けICH Q1A-Q1F安定性ガイドラインとの組み込み整合
FDA安定性要件コンプライアンス
21 CFR Part 211.166を含むFDA安定性要件に適合する事前設定ワークフロー
GMP安定性プログラム管理
継続的安定性コミットメントと承認後モニタリングを含む包括的なGMP安定性プログラム
安定性トレンド分析と有効期限予測
AIが安定性試験結果を分析し、製品劣化トレンドの特定と製品有効期限の推定を実施
Value Delivered
Key Benefits
Measurable outcomes that transform laboratory operations and deliver enterprise-wide value.
Automated Stability Protocol Creation
Eliminate manual protocol design with ICH Q1A-compliant templates that automate study setup, sampling schedules, and acceptance criteria for consistent, regulatory-ready stability protocol creation
Zero Missed Sample Pulls
Automated sample pull scheduling with calendar reminders, dynamic rescheduling, and escalation alerts ensures every pull point is executed on time with full audit trail
Centralized Chamber Management
Real-time chamber storage management with environmental monitoring, capacity planning, and excursion alerts provides complete visibility and control over all stability chambers
Faster Shelf-Life Determination
AI-powered shelf-life determination reduces product shelf-life evaluation time by 40% with statistical trend analysis, expiry determination, and confidence interval projections
Full ICH & FDA Compliance
Built-in compliance with ICH Q1A stability guidelines and FDA stability requirements ensures audit-ready documentation and regulatory submission readiness for GMP stability programs
Integrated Stability Trend Analysis
Automated stability trend analysis with degradation modeling, control charts, and statistical reporting enables data-driven decisions and accelerates regulatory submissions
Real-World Applications
Use Cases
Proven scenarios where this module delivers measurable value across laboratory and manufacturing operations.
Pharmaceutical Stability Studies — Small Molecule NDA/ANDA
Manage long-term, accelerated, and intermediate stability studies for NDA/ANDA submissions with automated stability protocol creation, sample pull scheduling, and AI-powered shelf-life determination aligned to ICH Q1A stability guidelines and FDA stability requirements
Reduce shelf-life determination time by 40% and enable faster product launches with regulatory-ready stability data packages
Biologics & Biosimilar Stability Programs
Coordinate complex multi-condition stability studies for protein therapeutics and biosimilars with real-time chamber storage management, environmental monitoring, and deviation tracking across multiple storage conditions
Maintain full ICH Q5C compliance while reducing analyst manual effort by 50% through automated stability program management
GMP Ongoing Stability Commitments
Manage post-approval ongoing GMP stability programs with automated annual stability assessment, commitment tracking, and trend monitoring to verify continued product quality throughout shelf-life
Ensure continuous GMP stability program compliance with zero missed commitments and proactive trend-based quality alerts
Contract Testing Labs — Multi-Client Stability Management
Operate as a stability testing management system for multiple sponsor clients with segregated studies, client-specific protocols, coordinated chamber allocation, and consolidated reporting
Increase concurrent study capacity by 3x with centralized stability study management software across all client programs
Common Questions
Frequently Asked Questions
Ready to Modernize 高度安定性管理?
See how AmpleLogic LIMS can transform your 高度安定性管理 workflows with a personalized demo.
ライフサイエンスの最前線へ
最新の製品アップデート、コンプライアンスニュース、業界インサイトをお届けします。