确保每位员工接受培训并符合 GxP 合规要求
面向生命科学的培训管理软件,具备 FDA cGMP 培训软件功能、自动化培训分配、GMP 培训追踪以及审计就绪培训管理——这是一款符合 21 CFR Part 11 的 LMS,专为受监管行业构建的云端 LMS。
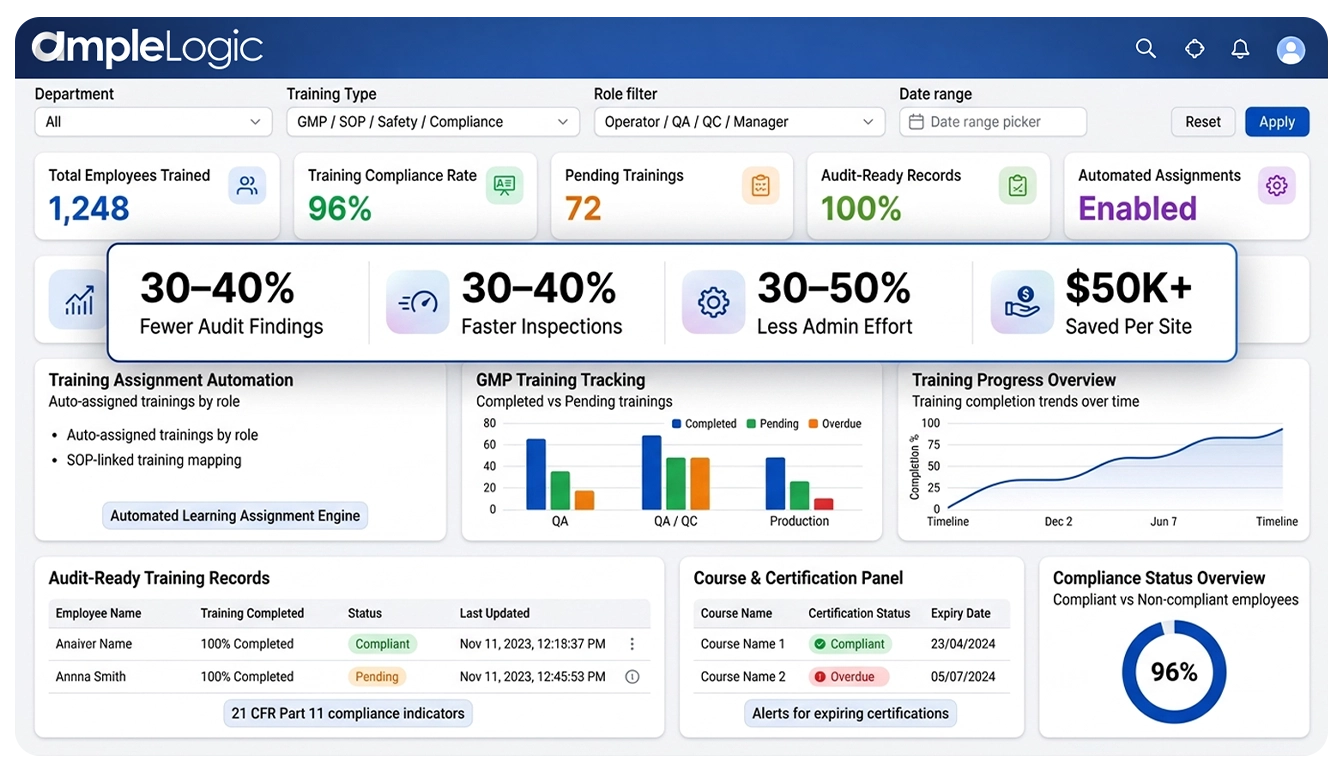
30-40%
审计发现减少
30-40%
检查速度提升
30-50%
管理工作量降低
$50K+
每站点节省成本
制药行业 GMP 培训管理的现状
手动系统和传统 LMS 平台造成合规漏洞、检查风险和运营效率低下
手动管理
员工培训管理依赖电子表格和纸质记录;分散、孤立,在 FDA 和 MHRA 检查期间存在高风险。
传统/部分集成的 LMS
受监管行业 LMS 部分自动化,集成有限,跨站点存在空白,GMP 培训追踪记录需手动核对。
手动合规每站点成本超过 $50K
碎片化系统是全球制药企业审计风险暴露和运营瓶颈的主要原因。
每次检查的人工准备时间超过 200 小时。
通过消除基于纸质的核对错误,每站点可避免超过 $50K 的整改成本。
碎片化数据孤岛因跨站点记录不一致而导致高检查风险。
制药 LMS 集成挑战:孤立系统如何制造合规风险
培训、质量和生产数据在制药 IT 体系中各自隔离——造成系统性合规风险
输入来源
ERP/HR
重复记录和角色
LMS
培训数据孤立
eQMS
SOP 更新未关联
RIMS
法规培训需求
断开连接的系统
平台间的手动交接
入职延迟
新员工等待数天至数周
沉重的审计负担
检查准备超过 200 小时
合规风险
执行后才发现培训空白
输出执行
LIMS
分析员资质未核查
MES
批次执行存在空白
CAPS
校准培训未强制执行
eLogbook
未核查设备认证
Cleaning Validation
清洁程序存在空白
面向制药行业的统一 SOP 培训架构
集中合规管理、自动化报告与实时能力可视化——集于单一经验证平台
集中化学习管理
跨全球运营的培训内容、交付和培训追踪软件功能的单一存储库——是面向制药行业的优质培训管理系统的基础。
基于角色的访问控制
细粒度权限确保人员仅访问所需培训和信息;支持符合 21 CFR Part 11 的 LMS 要求以及制药员工资质管理系统治理。
GMP 合规培训引擎
GMP 合规培训软件,具备自动化能力评估、法规要求映射和审计追踪生成功能,为 FDA 合规培训系统准备就绪。
实时报告仪表板
即时查看各站点完成情况、能力状态和质量合规培训指标——驱动检查准备培训平台的功能。
生物特征考勤
与生物特征设备无缝连接,在生产培训管理环境中实现防篡改的指纹认证考勤追踪。
企业集成
与 DMS、eQMS、ERP、HR 和 LIMS 集成,在此制药合规培训软件生态系统中实现端到端质量可追溯性。
集中化学习管理
跨全球运营的培训内容、交付和培训追踪软件功能的单一存储库——是面向制药行业的优质培训管理系统的基础。
基于角色的访问控制
细粒度权限确保人员仅访问所需培训和信息;支持符合 21 CFR Part 11 的 LMS 要求以及制药员工资质管理系统治理。
GMP 合规培训引擎
GMP 合规培训软件,具备自动化能力评估、法规要求映射和审计追踪生成功能,为 FDA 合规培训系统准备就绪。
实时报告仪表板
即时查看各站点完成情况、能力状态和质量合规培训指标——驱动检查准备培训平台的功能。
生物特征考勤
与生物特征设备无缝连接,在生产培训管理环境中实现防篡改的指纹认证考勤追踪。
企业集成
与 DMS、eQMS、ERP、HR 和 LIMS 集成,在此制药合规培训软件生态系统中实现端到端质量可追溯性。
AmpleLogic LMS 模块功能
从员工工作区到高管仪表板的全面培训生命周期管理
我的培训需求
您的个性化培训工作区
个性化仪表板,在此面向生命科学的培训管理软件中一目了然地显示已分配课程、即将到期时间、已完成的制药员工认证及能力状态。
主数据
集中化组织设置
员工详情
制药员工资质管理系统
培训手册
SOP 培训管理存储库
培训课程
多模式培训执行引擎
认证
制药员工认证与合规验证
指标
GMP 培训追踪与绩效监控
分析
AI 学习管理系统洞察
交互式仪表板
实时培训与合规可见性
现代、灵活的 GMP 学习体验
结合 AI 驱动的内容创建,旨在提升各角色的参与度、技能准备程度和培训效果
多种学习格式
支持基于文档的学习、录制培训、AI 生成播客(多语言)和在岗培训(OJT)——是受监管行业 LMS 中实现最大灵活性的综合解决方案。
基于 AI 的问卷生成
AI 学习管理系统可从培训内容自动创建题库,在保持评估质量和法规一致性的同时,将人工工作量减少高达 80%。
基于标准的学习支持
完全符合 SCORM 和 xAPI 标准,实现此制药质量培训软件中内容的无缝互操作性,并支持行业标准分析追踪。
个性化学习路径
基于角色、部门和职业阶段的学习路径,能够适应个人能力差距——提供可提升相关性和课程完成率的员工培训管理。
AI 生成播客
自动将培训材料转换为引人入胜的多语言音频内容,支持现场和生产培训管理人员随时随地学习。
互动评估
可配置的测验、情景评估和实操演示,具备自动评分和能力验证工作流,符合制药员工资质管理系统合规要求。
实时培训智能与绩效追踪
实时了解培训绩效、合规状态和劳动力准备就绪情况
合规与审计报告
通过完整的培训完成记录、能力文档和审计追踪摘要,即时生成符合监管要求的证据。
员工绩效与资质报告
通过详细的绩效分析和资质差距分析,确保每位员工接受培训并具备所分配角色的资质。
资质与认证追踪
通过自动到期通知和重新认证排期,在各站点维护经验证的认证记录。
课程规划与培训运营
通过需求预测、讲师利用率和课程效果指标,优化培训计划和资源规划。
构建审计就绪的培训系统
针对 FDA 21 CFR Part 11 和 GMP 审计准备设计培训,并自动生成证据
自动化审计追踪生成
符合 21 CFR Part 11 的 LMS
符合 21 CFR Part 11 的 LMS 记录培训、评估和能力记录,并附有时间戳和用户 ID,作为 FDA cGMP 培训软件实现完整的可追溯性。
实时合规报告
GMP 合规培训软件
GMP 合规培训软件仪表板实时显示各站点的培训状态、能力状态、到期情况和逾期事项,支持审计就绪培训管理。
内置 GMP 要求
FDA cGMP 培训软件
预配置的 GMP 模块和对齐的评估标准,作为制药质量培训软件——减少验证工作量,并借助 GMP 复训支持加速部署。
培训内容版本控制
SOP 培训管理更新
完整的内容变更历史,包含生效日期、审批记录,以及 SOP 修订时自动触发的重新培训——是 SOP 培训管理和质量合规培训的核心。
基于角色的访问控制
数据完整性与 GxP 培训软件
GxP 培训软件具备细粒度权限,确保仅授权人员访问敏感培训数据,在此 FDA 合规培训系统中支持 ALCOA+ 数据完整性。
检查准备培训平台
更快完成检查,减少审计发现
检查准备培训平台可在数分钟内响应检查员询问;审计时间减少 30-40%;通过审计就绪培训管理加快整改和证据呈现。
AI 驱动的预测性能力管理洞察
借助内置 AI 智能,从被动培训转向及时、以合规为重点的干预
预测能力
- AI 学习管理系统在技能差距影响实验室、生产或维护活动之前,提前识别新兴技能差距
- 根据劳动力变化、法规更新和年度 GMP 培训日历要求预测培训需求
风险与合规信号
- 在此 GxP 培训软件中自动检测异常培训和资质模式
- 标记缺少的前提条件、逾期的 GMP 复训或不合规的培训顺序
- 监控法规更新并推荐培训调整,以实现 FDA 合规培训系统对齐
运营与仪表板指标
- 培训追踪软件呈现培训效果和达到能力所需时间的趋势
- 培训矩阵软件按站点和角色呈现能力热图
- 检查准备培训平台指标和审计反馈趋势

AmpleLogic GMP 学习管理系统的业务影响与投资回报
使用最佳制药企业 LMS 的首席执行官和质量保证负责人可获得切实的合规、效率和成本保护收益
30-40%
培训相关审计发现减少
通过自动化培训分配、证据生成和此制药行业质量培训管理系统内的实时能力可见性,合规发现大幅下降。
30-40%
检查响应时间加快
通过审计就绪培训管理即时访问 GMP 培训追踪记录、审计追踪和资质文档,更快向检查员提供证据。
30-50%
培训管理工作量降低
通过自动化培训分配、排期、能力追踪和培训追踪软件报告工作流,培训团队的工作量减轻。
$50K+
每站点避免的整改成本
通过此面向受监管行业的云端 LMS 消除基于纸质的核对错误和手动培训空白,实现成本保护。
4-6 周
更快入职与质量提升
跨站点运营效益,以及实验室、生产和生产培训管理团队间一致的制药员工资质管理系统。
10 个月
完整投资回报周期
从碎片化到集成化:单一统一的生命科学培训管理软件平台,4-6 周内完成部署,10 个月内实现完整投资回报。
面向生命科学的 aPaaS——受监管行业的云端 LMS
通过制药合规培训软件在整个企业生态系统中实现端到端资质强制执行
DMS
inbound来自新 SOP 引入和版本变更的培训需求——在文档更新时自动触发重新培训。
eQMS
inbound来自变更控制、审计发现、偏差和 OOS/OOT 事件的培训需求,用于纠正措施。
RIMS
inbound来自法规发现的培训需求——自动将法规要求映射到培训课程。
LIMS
outbound实时分析员资质验证——确保仅受过培训的分析员执行实验室程序。
eLog Book
outbound设备访问控制——在授予设备登录访问权限之前验证培训认证。
Cleaning Validation
outbound清洁程序的受训人员强制执行——阻止未受过培训的操作员执行协议。
eBMR / MES
outbound操作员认证强制执行——在批次执行之前验证生产人员资质。
CAPS
outbound校准培训强制执行——确保技术员在执行校准活动之前接受培训。
Quality Metrics
outbound培训效果数据流入质量仪表板——将培训完成情况与质量 KPI 关联。
SAP / ERP
双向双向员工数据同步、成本中心对齐和组织层级管理。
HR Systems
双向员工入职触发、角色变更和离职工作流自动更新培训分配。
UMS
双向统一用户管理,包含单点登录、访问权限配置以及跨所有模块的身份治理。
核心供应商差异化优势
AmpleLogic 制药质量培训软件有别于传统和旧式学习管理平台之处
最深度的 aPaaS 集成
与仅限 QMS 集成的竞争对手不同,AmpleLogic 将此 GMP 学习管理系统与 14 个以上模块(包括 LIMS、eBMR/MES、CAPS 和 Cleaning Validation)原生连接——提供端到端资质强制执行。
AI 原生智能
内置 AI 学习管理系统代理,用于问卷生成、多语言播客创建、预测性能力管理和自动化培训差距分析——超越制药合规培训软件中的基础自动化。
资质强制执行
跨生产培训管理(MES)、实验室培训管理(LIMS)、校准(CAPS)和清洁操作实时阻止不合格人员——这是竞争对手在执行点无法提供的能力。
快速实现价值
4-6 周内完成实施,提供预配置的 GMP 模块、经验证的模板和符合 GAMP 5 的文档——而传统受监管行业 LMS 供应商的典型周期为 6-12 个月。
我们服务的行业
面向生命科学和受监管行业的 GxP 合规培训解决方案
制药
面向制药企业的最佳 LMS,为生产和质量团队提供符合 GMP 的培训
生物技术
GxP 培训软件,为研究和生产提供复杂的资质认定路径
医疗器械
面向器械公司的符合 ISO 13485 的培训和员工培训管理
CRO / CDMO
具备审计就绪培训管理记录的多客户培训隔离
生命科学
面向生命科学的培训管理软件,提供跨全球站点的企业级学习治理
制造业
具备操作员认证和设备资质追踪的生产培训管理
借助实时培训智能驱动更明智的决策
加入领先的生命科学机构,使用 AmpleLogic——制药企业的最佳 LMS——加速质量合规培训、降低审计风险,并借助面向生命科学的培训管理软件构建以能力为导向的文化。
引领生命科学前沿
获取最新产品更新、合规动态及行业洞察,直接发送至您的邮箱。