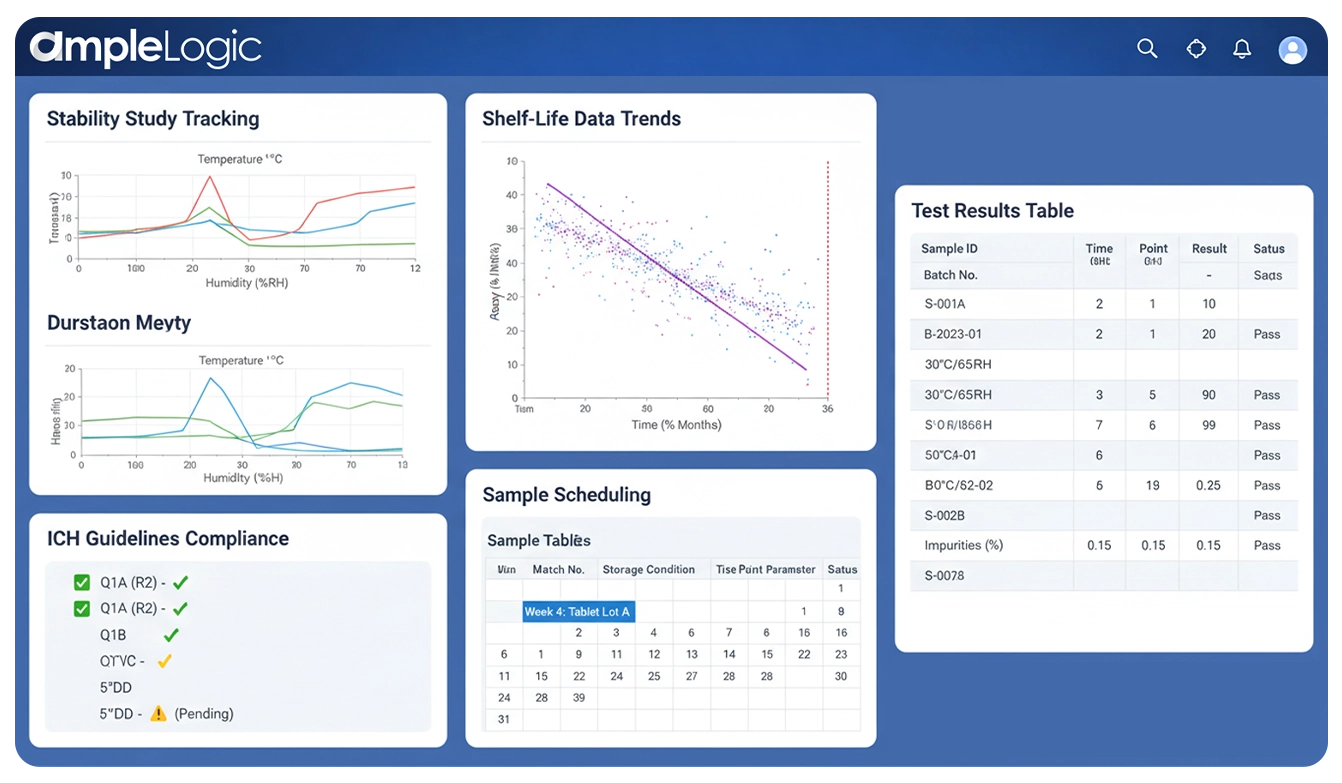
在LIMS中端到端管理稳定性研究
AmpleLogic提供端到端的稳定性计划管理——从稳定性试验方案创建和稳定性计划日历规划,到챔버环境监测、试验执行、趋势分析、货架期确定,以及符合ICH Q1A、FDA和GMP要求的法规合规报告。
40%
研究设置效率提升
100%
ICH 合规性
30%
减少取样点遗漏
60%
减少手动追踪工作
50%
成本节约
3–6
个月部署周期
什么是稳定性计划管理?
稳定性计划管理是对药品稳定性研究进行系统性规划、执行和追踪的过程。它确保每个产品批次按照稳定性试验方案的规定,在正确的储存条件、取样时间点和分析检测要求下进行,从而实现准确的货架期确定,并符合ICH Q1A、FDA 21 CFR Part 211.166和GMP法规要求。
稳定性试验方案
稳定性试验方案定义了研究设计——储存条件、时间点、检测参数、验收标准和包装配置。AmpleLogic通过ICH Q1A合规模板自动化方案创建流程。
稳定性计划日历
稳定性计划日历是所有进行中研究的取样日期、챔버分配和分析人员安排的可视化时间表。AmpleLogic实时自动生成并维护该日历。
稳定性计划管理
稳定性计划管理在单一经验证的平台中协调整个生命周期——从方案审批到样品取样、试验执行、趋势分析和货架期确定。
缺乏自动化的稳定性计划管理所面临的挑战
依赖手动流程进行稳定性试验方案管理和稳定性计划日历追踪的组织面临严重风险。
手动稳定性计划管理
团队依赖电子表格和纸质记录规划稳定性计划,导致取样时间点遗漏、计划冲突,以及对研究时间表缺乏实时可见性。
稳定性试验方案不一致
缺乏标准化系统,稳定性试验方案的创建因团队和站点而异,导致法规缺口、审计发现问题和研究设计不一致。
缺乏集中化的稳定性计划日历
缺少统一的稳定性计划日历,分析人员无法在一处查看即将到来的取样、챔버分配或重叠研究情况。
环境数据相互脱节
챔버温湿度数据与稳定性研究分开追踪,造成可追溯性缺口和偏差响应延迟。
趋势分析和报告延迟
手动数据汇编用于稳定性趋势分析,延误货架期确定、法规申报和产品上市时间表。
分散系统带来的合规风险
使用多个分散工具进行稳定性项目管理会增加数据完整性风险,使ICH Q1A和FDA审计准备工作变得困难。
AmpleLogic稳定性试验方案和计划自动化的实施方法
六个集成步骤,将手动稳定性计划管理转变为完全自动化的ICH合规工作流程。
定义稳定性试验方案
在单一经验证的系统中,创建包含可配置研究设计、储存条件、验收标准和自动取样计划的ICH Q1A合规稳定性试验方案。
构建稳定性计划日历
自动生成包含时间点取样日期、챔버分配、分析人员配置和自动提醒的可视化稳定性计划日历——完全消除手动计划工作。
监测챔버条件
实时温湿度监测,具有自动偏差警报、传感器集成,以及与稳定性研究记录直接关联的环境数据。
执行试验并记录结果
在集成LIMS工作流程中运行分析试验,具备直接仪器连接、内联计算和完整的样品到结果可追溯性。
分析趋势并确定货架期
AI驱动的稳定性趋势分析,包含降解建模、统计货架期预测、有效期确定和置信区间报告。
生成法规报告
从稳定性数据中自动生成ICH合规稳定性报告、FDA申报包和GMP稳定性项目文档。
AmpleLogic稳定性计划日历及数据中心
集稳定性试验方案管理、稳定性计划日历、趋势分析和合规文档于一体的集中化平台。
统一研究仪表板
涵盖所有产品和条件的所有进行中、已完成和计划中稳定性研究的单一视图,包含实时状态、即将到来的取样日期和逾期警报。
稳定性计划管理引擎
集中化稳定性计划管理,具备自动取样排程、动态重新计划、基于优先级的队列管理和每项研究的日历视图。
交互式趋势分析
多批次叠加图表、降解曲线拟合、OOT/OOS检测和预测性货架期预测——所有功能均可从单一分析控制台访问。
完整审计追踪
从稳定性试验方案创建到结果录入和报告生成的每项操作,均以带时间戳、用户标识、符合ALCOA+合规要求的审计记录进行捕获。
智能通知中心
针对即将到来的取样、逾期试验、OOT结果、챔버偏差和方案偏差的可配置警报,以及多级升级工作流程。
跨系统集成
与LIMS、ERP、QMS和仪器系统的双向集成,为组织内所有稳定性数据建立单一可信数据源。
手动 vs. 自动化稳定性计划管理
了解AmpleLogic如何全面改变稳定性试验方案管理和稳定性计划日历运营的各个方面。
| 领域 | 实施前(手动) | 实施后(AmpleLogic) |
|---|---|---|
| 研究排程 | 手动电子表格追踪、计划冲突 | 带提醒的自动化稳定性计划日历 |
| 方案创建 | Word文档,各站点格式不统一 | 标准化稳定性试验方案模板(ICH Q1A) |
| 取样管理 | 取样遗漏,无实时可见性 | 自动排程和警报实现零遗漏 |
| 챔버监测 | 传感器脱节,偏差响应延迟 | 具备即时警报的实时监测 |
| 趋势分析 | 手动Excel图表,汇编需数周 | AI驱动分析,即时货架期预测 |
| 报告 | 手动汇编,易发生错误 | 一键生成ICH/FDA法规合规报告 |
| 审计准备 | 仓促收集证据 | 完整ALCOA+追踪,始终保持审计就绪状态 |
生命科学领域广泛信赖
稳定性试验方案和计划管理的法规合规性
引领生命科学前沿
获取最新产品更新、合规动态及行业洞察,直接发送至您的邮箱。